1
answer
0
watching
236
views
13 Dec 2019
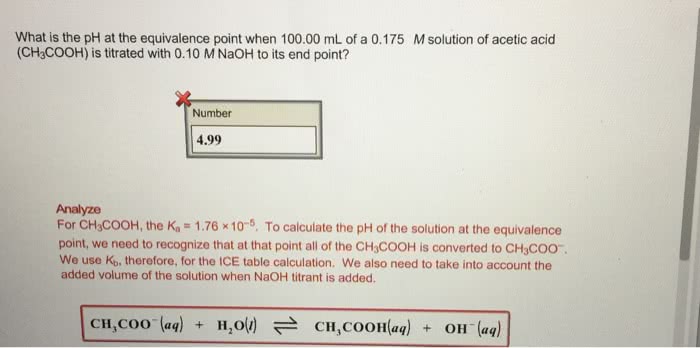
What is the pH at the equivalenve point when 100.00 mL of a 0.175 M solution of acetic acid (CH3COOH) is titrated with 0.10 M NaOH to its end point?
What is the pH at the equivalenve point when 100.00 mL of a 0.175 M solution of acetic acid (CH3COOH) is titrated with 0.10 M NaOH to its end point?
Beverley SmithLv2
17 Dec 2019