1
answer
0
watching
613
views
13 Dec 2019
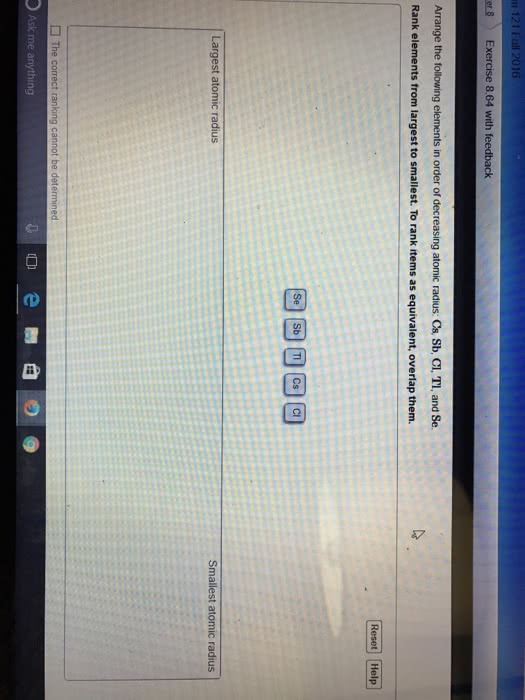
Each element in the periodic table has a distinctive atomic radius.
Place the following elements in order of decreasing atomic size: antimony, selenium, fluorine, rubidium, strontium, and arsenic.
Rank from largest to smallest. To rank items as equivalent, overlap them.
F, Se, Rb, As, Sr, Sb
Each element in the periodic table has a distinctive atomic radius.
Place the following elements in order of decreasing atomic size: antimony, selenium, fluorine, rubidium, strontium, and arsenic.
Rank from largest to smallest. To rank items as equivalent, overlap them.
F, Se, Rb, As, Sr, Sb
Sixta KovacekLv2
17 Dec 2019