0
answers
0
watching
238
views
13 Dec 2019
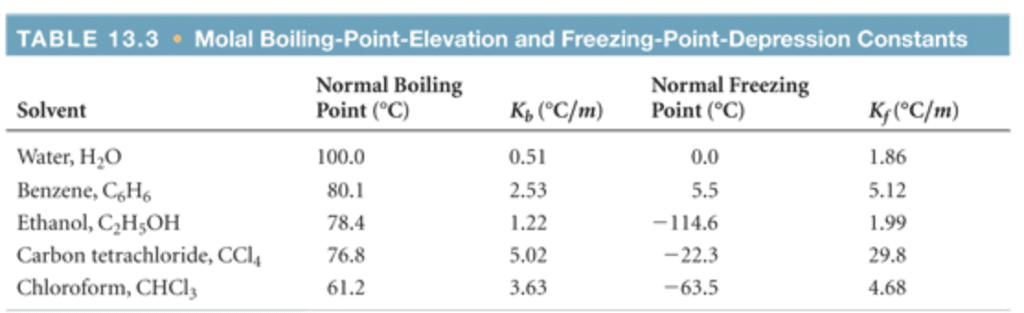
Boiling Point Elevation/Freezing Point Depression T = m K Where: T = T(solution) - T(pure solvent) * m = (# moles solute / Kg solvent) Kb = boiling point elevation constant. Kf = freezing point depression constant. Kb and Kf depend only on the SOLVENT. Below are some common values. Use these values for the calculations that follow.
Solvent Formula Kb(c/m) Kr(c/m)
water H2O 0.512 -1.86
Ethanol CH3CH2OH 1.22 -1.99
Chloroform CHCl3
Benzene C6H6 2.53 -5.12
Diethyl ether CH3CH2OCH2CH3
*Please note that ÎT as defined above will be a negative number for freezing point depression. Therefore, Kf must also be given as a negative number
Boiling Point Elevation/Freezing Point Depression T = m K Where: T = T(solution) - T(pure solvent) * m = (# moles solute / Kg solvent) Kb = boiling point elevation constant. Kf = freezing point depression constant. Kb and Kf depend only on the SOLVENT. Below are some common values. Use these values for the calculations that follow.
Solvent Formula Kb(c/m) Kr(c/m)
water H2O 0.512 -1.86
Ethanol CH3CH2OH 1.22 -1.99
Chloroform CHCl3
Benzene C6H6 2.53 -5.12
Diethyl ether CH3CH2OCH2CH3
*Please note that ÎT as defined above will be a negative number for freezing point depression. Therefore, Kf must also be given as a negative number