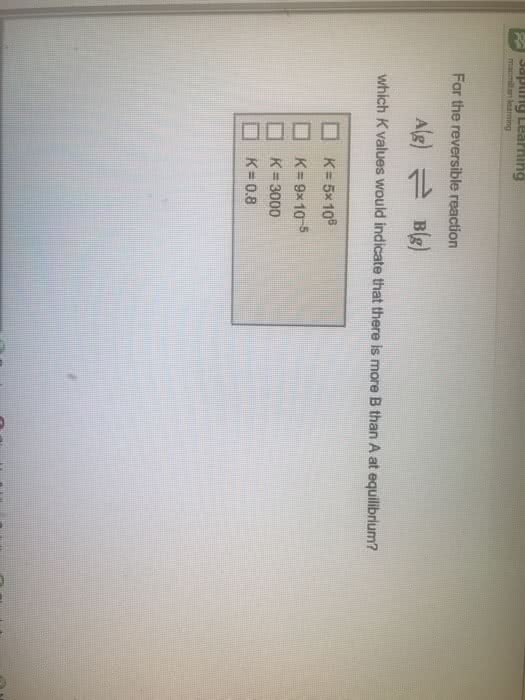
1
answer
0
watching
135
views
13 Dec 2019
Which of the following statements are CORRECT? 1. For gas phase equilibria, the partial pressures of reactants and products are equal. 2. For a chemical system at equilibrium, the forward and reverse rates of reaction are equal. 3. For an aqueous chemical system at equilibrium, the concentrations of products divided by the concentrations of reactants equals one.
Which of the following statements are CORRECT? 1. For gas phase equilibria, the partial pressures of reactants and products are equal. 2. For a chemical system at equilibrium, the forward and reverse rates of reaction are equal. 3. For an aqueous chemical system at equilibrium, the concentrations of products divided by the concentrations of reactants equals one.
Sixta KovacekLv2
17 Dec 2019