1
answer
0
watching
1,212
views
13 Dec 2019
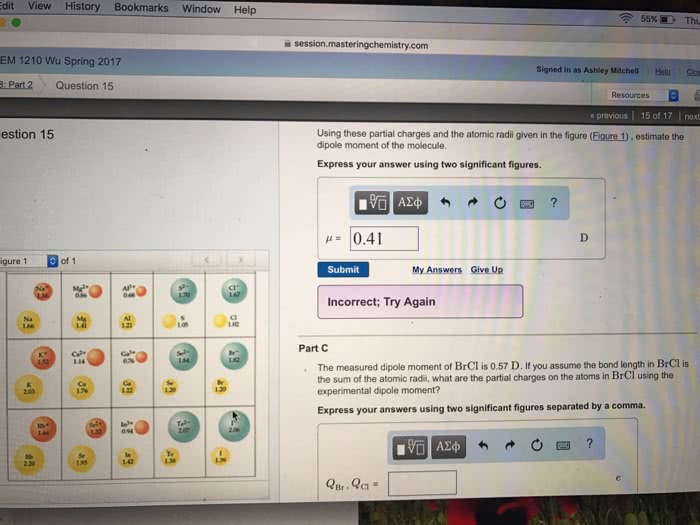
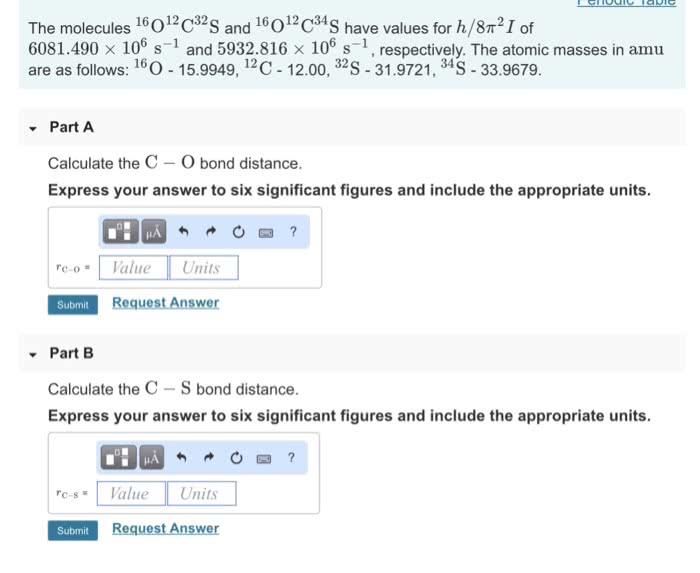
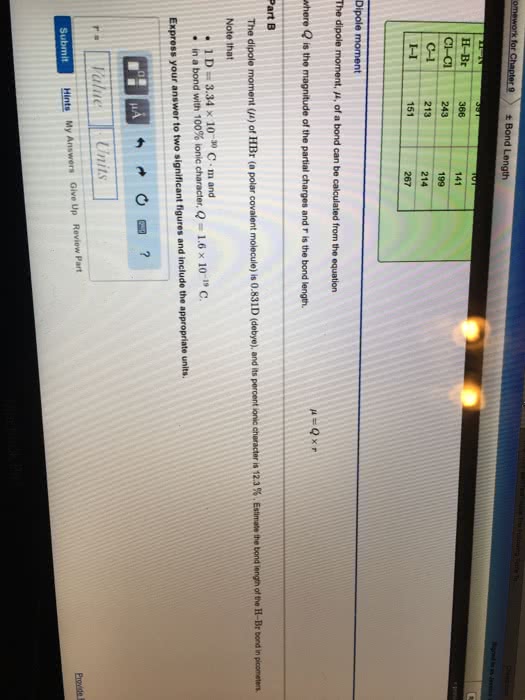
Part C Bond length is the distance between the centers of two bonded atoms. On the potential energy curve, the bond length is the internuclear distance between the two atoms when the potential energy of the system reaches its lowest value. Given that the atomic radii of H and Cl are 25.0 pm and 100 pm , respectively, predict the bond length of the HCl molecule. Express your answer to three significant figures and include the appropriate units.
Part C Bond length is the distance between the centers of two bonded atoms. On the potential energy curve, the bond length is the internuclear distance between the two atoms when the potential energy of the system reaches its lowest value. Given that the atomic radii of H and Cl are 25.0 pm and 100 pm , respectively, predict the bond length of the HCl molecule. Express your answer to three significant figures and include the appropriate units.
Beverley SmithLv2
17 Dec 2019