1
answer
0
watching
180
views
13 Dec 2019
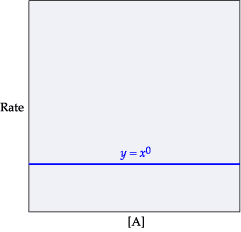
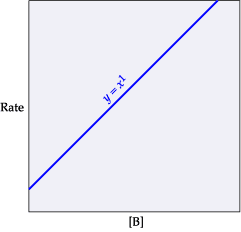
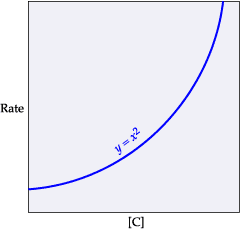
The rate of reaction in terms of the "rate law expression" includes the rate constant (k), the concentration of the reactants, and the orders of the reaction with respect to the different reactants. Consider the following reaction: A+BâC+D The initial concentrations of the reactants A and B are 0.240 M and 0.400 M, respectively. The rate of reaction is 0.060 Mâ sâ1, and the orders of the reaction, with respect to reactants A and B, are 1 and 2, respectively. Determine the rate constant (k) for the reaction using the rate law. Express your answer in Mâ2â sâ1 to three significant figures.
The rate of reaction in terms of the "rate law expression" includes the rate constant (k), the concentration of the reactants, and the orders of the reaction with respect to the different reactants. Consider the following reaction: A+BâC+D The initial concentrations of the reactants A and B are 0.240 M and 0.400 M, respectively. The rate of reaction is 0.060 Mâ sâ1, and the orders of the reaction, with respect to reactants A and B, are 1 and 2, respectively. Determine the rate constant (k) for the reaction using the rate law. Express your answer in Mâ2â sâ1 to three significant figures.
Deanna HettingerLv2
17 Dec 2019