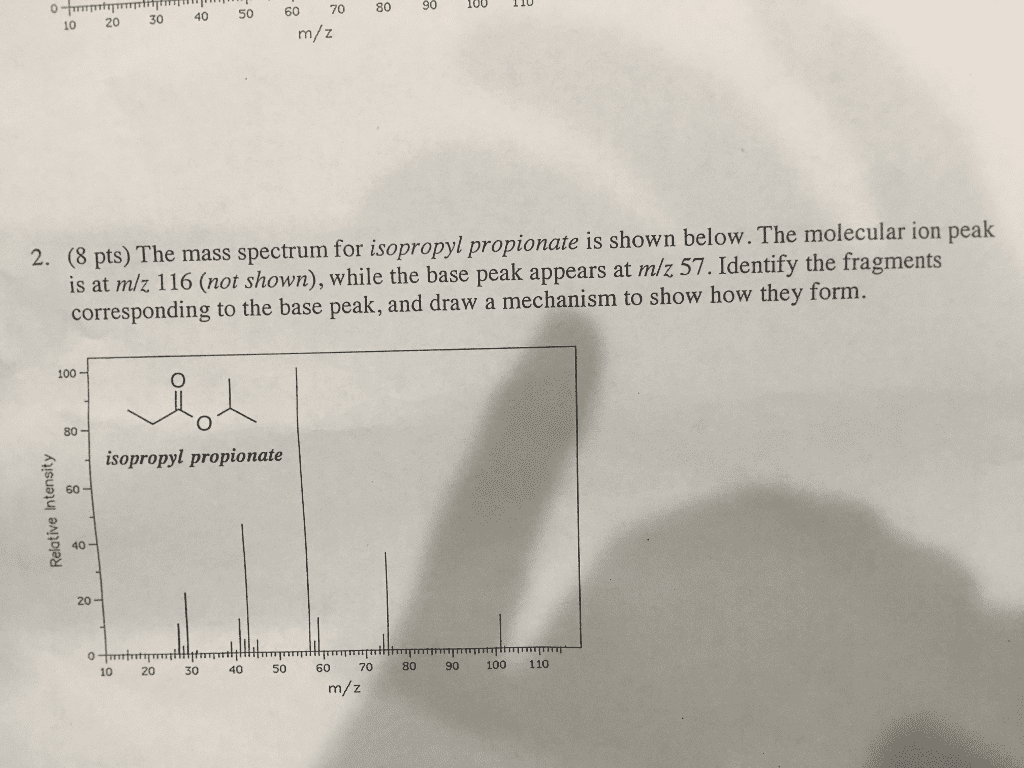1
answer
1
watching
639
views
13 Dec 2019
The mass of spectrum of n-octane shows a prominent molecular ion peak (m/z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethyhexane shows a smaller molecular ion, and the peak at mass 57 is the base peak. Explain these trends in abundance of the molecular ions and the ions at mass 57, and predict the intensities of the peaks at masses 57 and 114 in the spectrum of 2,2,3,3-tetramethylbutane.
The mass of spectrum of n-octane shows a prominent molecular ion peak (m/z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethyhexane shows a smaller molecular ion, and the peak at mass 57 is the base peak. Explain these trends in abundance of the molecular ions and the ions at mass 57, and predict the intensities of the peaks at masses 57 and 114 in the spectrum of 2,2,3,3-tetramethylbutane.
Liked by maryamzulfiqar075
Casey DurganLv2
17 Dec 2019