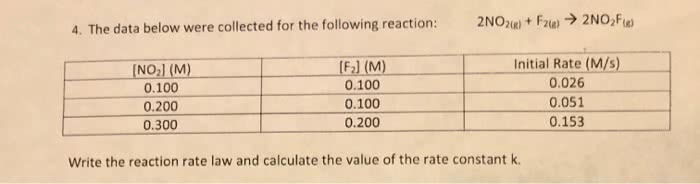1
answer
0
watching
223
views
13 Dec 2019
For the following combination reaction,
2 NO2(g) + F2(g) = 2 NO2F(g)
The initial rates of this reaction were determined for several concentrations of the reactants. All of the data is summarized via the following table:
Experiment [NO2] (M) [F2] (M) Rate (M/s) 1 0.100 0.100 0.026 2 0.200 0.100 0.051 3 0.200 0.200 0.103 4 0.400 0.400 0.411
a) Determine the rate law for this reaction based on the data above.
b) Calculate the rate constant, with appropriate units.
For the following combination reaction,
2 NO2(g) + F2(g) = 2 NO2F(g)
The initial rates of this reaction were determined for several concentrations of the reactants. All of the data is summarized via the following table:
| Experiment | [NO2] (M) | [F2] (M) | Rate (M/s) |
| 1 | 0.100 | 0.100 | 0.026 |
| 2 | 0.200 | 0.100 | 0.051 |
| 3 | 0.200 | 0.200 | 0.103 |
| 4 | 0.400 | 0.400 | 0.411 |
a) Determine the rate law for this reaction based on the data above.
b) Calculate the rate constant, with appropriate units.
Elin HesselLv2
17 Dec 2019