1
answer
0
watching
133
views
13 Dec 2019
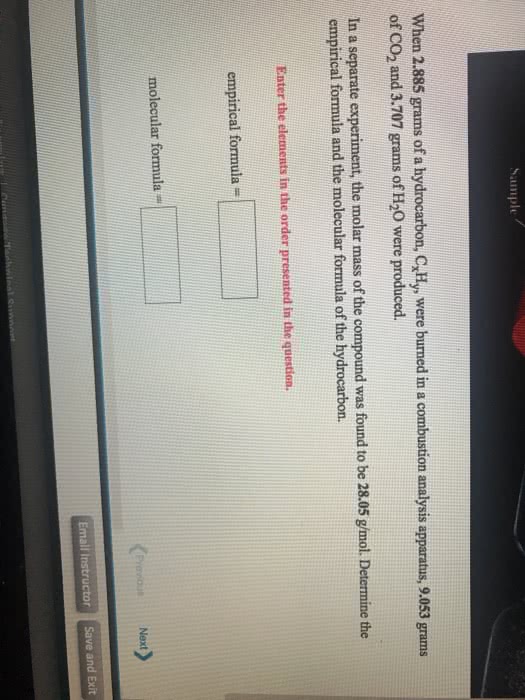
When 5.896 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 18.07 grams of CO2 and 8.630 grams of H2O were produced.
In a separate experiment, the molar mass of the compound was found to be 86.18 g/mol.
Determine the empirical formula and the molecular formula of the hydrocarbon.
When 5.896 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 18.07 grams of CO2 and 8.630 grams of H2O were produced.
In a separate experiment, the molar mass of the compound was found to be 86.18 g/mol.
Determine the empirical formula and the molecular formula of the hydrocarbon.
Keith LeannonLv2
17 Dec 2019