1
answer
0
watching
125
views
13 Dec 2019
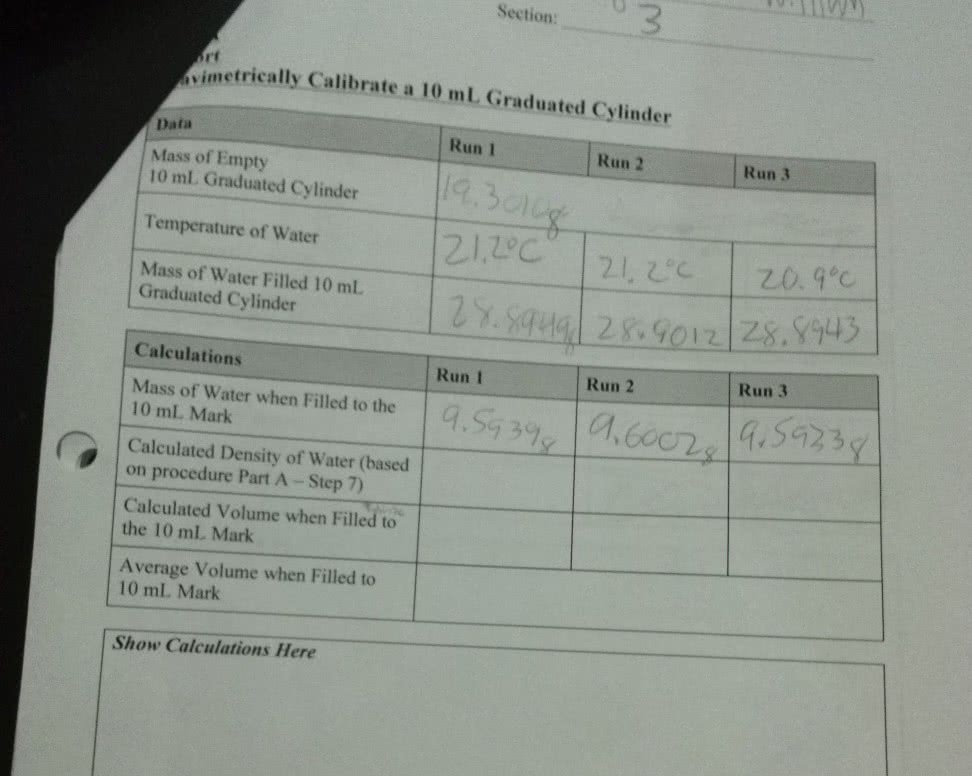
A student determines the mass of a 10 ml graduated cylinder to be 23.807 g. This same cylinder weighs 33.584 g when filled to the 10 ml calibration mark on this cylinder with the water at 22 degrees C. The density of the water at 22 degrees C is 0.9977g/ml. A) calculate the mass of water held in the cylinder. B) based on this data, what is the accurate volume of this cylinder when. filled up to the 10 ml mark at 22 degrees C? show work to defend answer. C) what is the percent error for the cylinder?
A student determines the mass of a 10 ml graduated cylinder to be 23.807 g. This same cylinder weighs 33.584 g when filled to the 10 ml calibration mark on this cylinder with the water at 22 degrees C. The density of the water at 22 degrees C is 0.9977g/ml.
A) calculate the mass of water held in the cylinder.
B) based on this data, what is the accurate volume of this cylinder when. filled up to the 10 ml mark at 22 degrees C? show work to defend answer.
C) what is the percent error for the cylinder?
Tod ThielLv2
17 Dec 2019