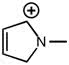
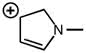
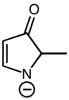
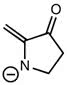
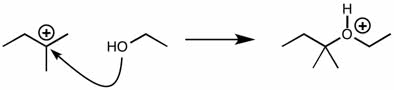
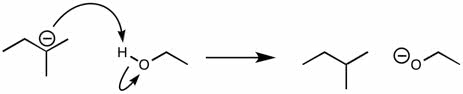
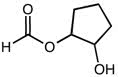
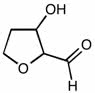
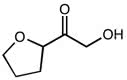
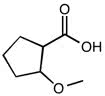
2
answers
0
watching
422
views
13 Dec 2019
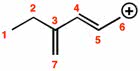
Which of the following statements is not true regarding molecular orbital theory?
a.) Molecular orbitals are regions of a molecule where the electrons are most likely to be found.
b.) Bonding molecular orbitals have lower energy than the atomic orbitals from which they are formed.
c.) Bonding molecular orbitals have lower energy than the atomic orbitals from which they are formed.
d.) Molecular orbitals are formed by combination of atomic orbitals.
Which of the following statements is not true regarding molecular orbital theory?
a.) Molecular orbitals are regions of a molecule where the electrons are most likely to be found.
b.) Bonding molecular orbitals have lower energy than the atomic orbitals from which they are formed.
c.) Bonding molecular orbitals have lower energy than the atomic orbitals from which they are formed.
d.) Molecular orbitals are formed by combination of atomic orbitals.
abdulmouizLv10
24 Apr 2023
Tod ThielLv2
17 Dec 2019
Already have an account? Log in