1
answer
0
watching
273
views
13 Dec 2019
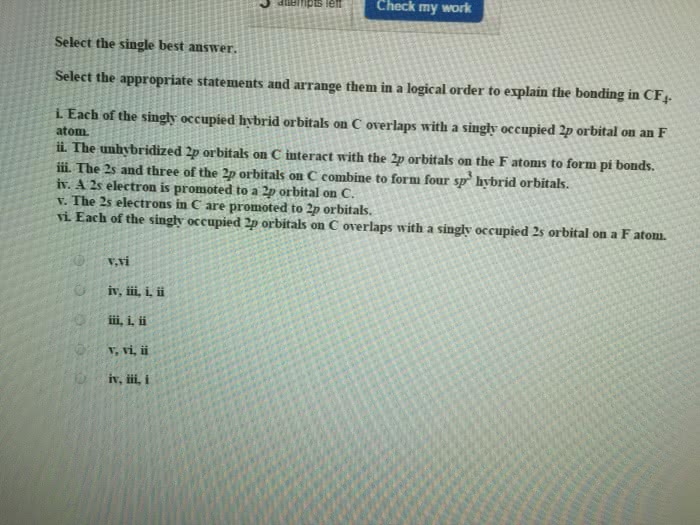
Which statement best describes what happens to the carbon atom in formaldehyde as a result of the hybridization process?
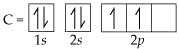
The electrons in the neutral carbon atom are redistributed so that there are three lone electrons in the three 2p orbitals and one lone electron in the 2s orbital. Hybridization of carbon results in the breakup of an electron pair and a slight increase in the overall energy of the system. The geometry of the sp2 hybrids points at the corners of an equilateral triangle. Carbon
|
1
answer
0
watching
273
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Collen VonLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232