1
answer
0
watching
80
views
13 Dec 2019
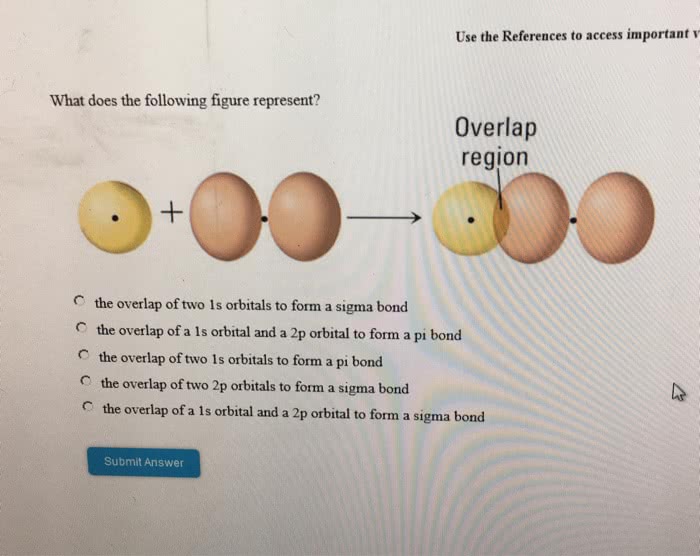
Which of the following statements is/are CORRECT according tovalence bond theory?
1.
The overlap between an s orbital and a porbital is called a pi-bond.
2.
The overlap of two s orbitals in H2 iscalled a sigma bond.
3.
HF is formed from the overlap of a hydrogen 1s orbitalwith a fluorine 2p orbital.
a.
1 only
b.
2 only
c.
3 only
d.
2 and 3
e.
1, 2, and 3
Which of the following statements is/are CORRECT according tovalence bond theory?
1. | The overlap between an s orbital and a porbital is called a pi-bond. | |||||||||||
2. | The overlap of two s orbitals in H2 iscalled a sigma bond. | |||||||||||
3. | HF is formed from the overlap of a hydrogen 1s orbitalwith a fluorine 2p orbital.
|
Beverley SmithLv2
17 Dec 2019