0
answers
0
watching
221
views
13 Dec 2019
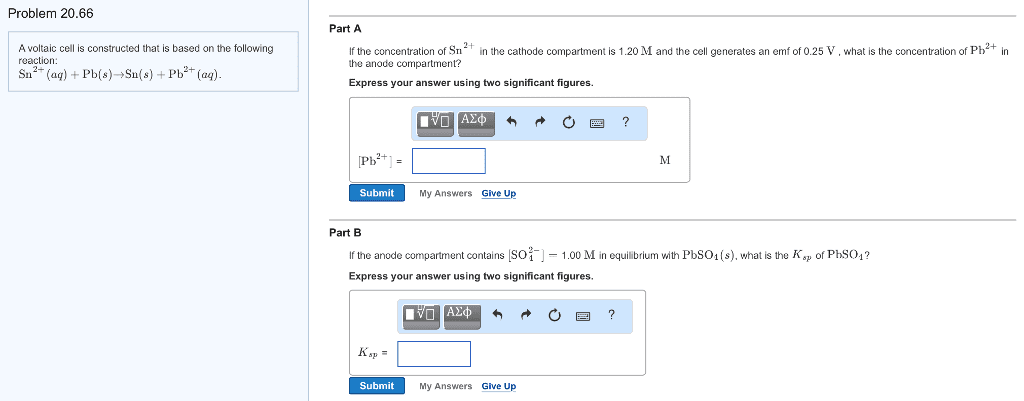
A voltaic cell is constructed that is based onthe following reaction:
Sn2+(aq)+Pb(s)?Sn(s)+Pb2+(aq).
Part A
If the concentration of Sn2+ in the cathodecompartment is 1.00M and the cell generates an emf of 0.20V , whatis the concentration of Pb2+ in the anode compartment?
Express your answer using two significantfigures.
Part B
If the anode compartment contains [SO2?4]=1.40M in equilibrium with PbSO4(s), what is the Ksp of PbSO4?
Express your answer using two significantfigures.
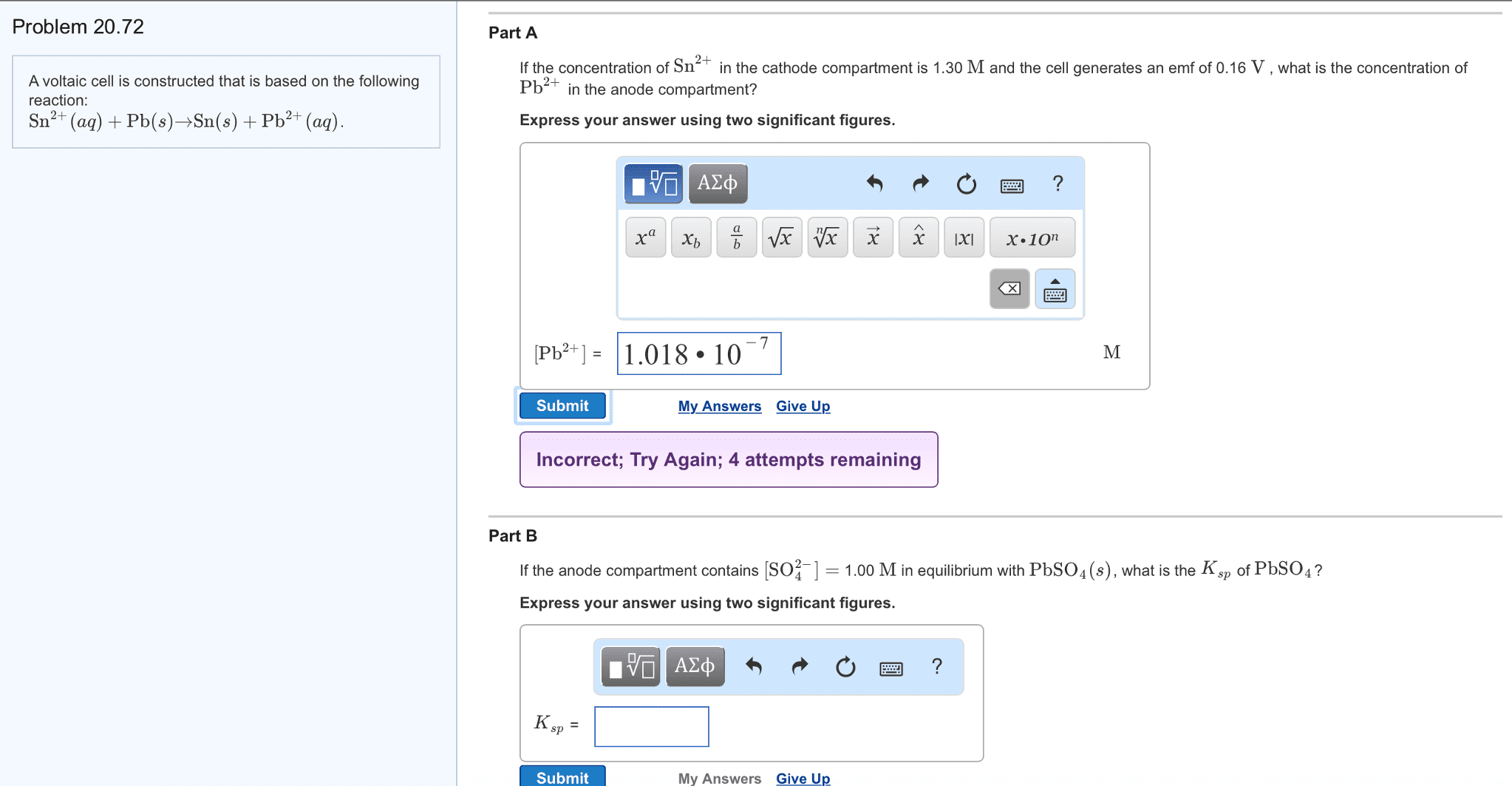
A voltaic cell is constructed that is based onthe following reaction:
Sn2+(aq)+Pb(s)?Sn(s)+Pb2+(aq).
Part A
If the concentration of Sn2+ in the cathodecompartment is 1.00M and the cell generates an emf of 0.20V , whatis the concentration of Pb2+ in the anode compartment?
Express your answer using two significantfigures.
Part B
If the anode compartment contains [SO2?4]=1.40M in equilibrium with PbSO4(s), what is the Ksp of PbSO4?
Express your answer using two significantfigures.