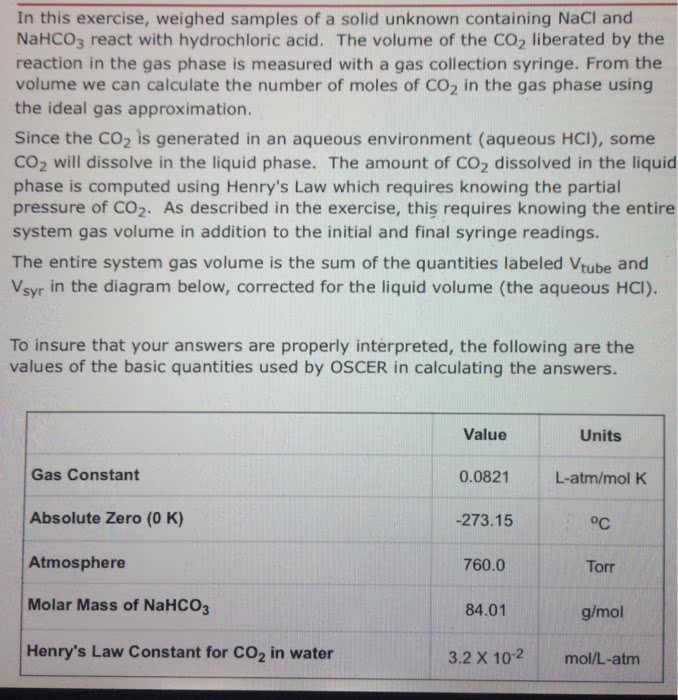
In this exercise, weighed samples of a solid unknown containing NaCl and NaHCO3react with hydrochloric acid. The volume of the CO2 liberated by the reaction in the gas phase is measured with a gas collection syringe. From the volume we can calculate the number of moles of CO2 in the gas phase using the ideal gas approximation.
Since the CO2 is generated in an aqueous environment (aqueous HCl), some CO2will dissolve in the liquid phase. The amount of CO2 dissolved in the liquid phase is computed using Henry's Law which requires knowing the partial pressure of CO2. As described in the exercise, this requires knowing the entire system gas volume in addition to the initial and final syringe readings.
The entire system gas volume is the sum of the quantities labeled Vtube and Vsyr in the diagram below, corrected for the liquid volume (the aqueous HCl).
Value Units Gas Constant 0.0821 L-atm/mol K Absolute Zero (0 K) -273.15 oC Atmosphere 760.0 Torr Molar Mass of NaHCO3 84.01 g/mol Henry's Law Constant for CO2in water 3.2 X 10-2 mol/L-atm
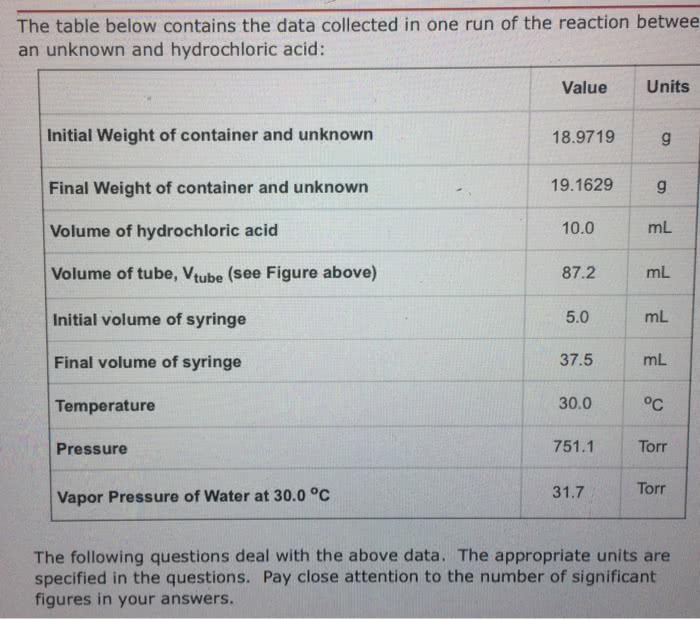
The table below contains the data collected in one run of the reaction between an unknown and hydrochloric acid:
Value Units Initial Weight of container and unknown
18.4395 g Final Weight of container and unknown 18.6185 g Volume of hydrochloric acid 10.0 mL Volume of tube, Vtube (see Figure above) 92.3 mL Initial volume of syringe 5.0 mL Final volume of syringe 50.7 mL Temperature 26.5 oC Pressure 764.4 Torr Vapor Pressure of Water at 26.5 oC 25.8 Torr
The following questions deal with the above data. The appropriate units are specified in the questions. Pay close attention to the number of significant figures in your answers.
1). What is the volume of CO2 captured in the gas phase (in mL)?
2).What is the number of mmols of CO2 captured in the gas phase? (Remember to account for the vapor pressure of water.)
3). What is the weight of the sample in grams?
In this exercise, weighed samples of a solid unknown containing NaCl and NaHCO3react with hydrochloric acid. The volume of the CO2 liberated by the reaction in the gas phase is measured with a gas collection syringe. From the volume we can calculate the number of moles of CO2 in the gas phase using the ideal gas approximation.
Since the CO2 is generated in an aqueous environment (aqueous HCl), some CO2will dissolve in the liquid phase. The amount of CO2 dissolved in the liquid phase is computed using Henry's Law which requires knowing the partial pressure of CO2. As described in the exercise, this requires knowing the entire system gas volume in addition to the initial and final syringe readings.
The entire system gas volume is the sum of the quantities labeled Vtube and Vsyr in the diagram below, corrected for the liquid volume (the aqueous HCl).
| Value | Units | |
| Gas Constant | 0.0821 | L-atm/mol K |
| Absolute Zero (0 K) | -273.15 | oC |
| Atmosphere | 760.0 | Torr |
| Molar Mass of NaHCO3 | 84.01 | g/mol |
| Henry's Law Constant for CO2in water | 3.2 X 10-2 | mol/L-atm |
The table below contains the data collected in one run of the reaction between an unknown and hydrochloric acid:
| Value | Units | |
| Initial Weight of container and unknown | 18.4395 | g |
| Final Weight of container and unknown | 18.6185 | g |
| Volume of hydrochloric acid | 10.0 | mL |
| Volume of tube, Vtube (see Figure above) | 92.3 | mL |
| Initial volume of syringe | 5.0 | mL |
| Final volume of syringe | 50.7 | mL |
| Temperature | 26.5 | oC |
| Pressure | 764.4 | Torr |
| Vapor Pressure of Water at 26.5 oC | 25.8 | Torr |
The following questions deal with the above data. The appropriate units are specified in the questions. Pay close attention to the number of significant figures in your answers.
1). What is the volume of CO2 captured in the gas phase (in mL)?
2).What is the number of mmols of CO2 captured in the gas phase? (Remember to account for the vapor pressure of water.)
3). What is the weight of the sample in grams?