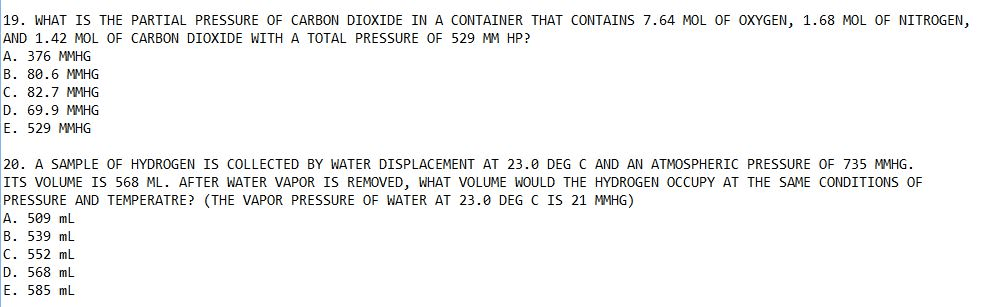
1
answer
0
watching
586
views
13 Dec 2019
Aqueous solutions of NH3 and HCN react to produce NH4CN according to the following equilibrium reaction.
NH3(aq) +HCN(aq) <---> NH4+(aq) +CN-(aq)
Given the following equilibrium constants, which statement best describes the reaction once equilibrium is established? (Kw=1.01x10^-14) and why?
NH4+ Ka = 5.6x10^-10
HCN Ka=4.0x10^-10
A. The reaction is running in forward direction.
B. The reaction is running in reverse direction
C. The reaction is neither product nor reactant favored
Aqueous solutions of NH3 and HCN react to produce NH4CN according to the following equilibrium reaction.
NH3(aq) +HCN(aq) <---> NH4+(aq) +CN-(aq)
Given the following equilibrium constants, which statement best describes the reaction once equilibrium is established? (Kw=1.01x10^-14) and why?
NH4+ Ka = 5.6x10^-10
HCN Ka=4.0x10^-10
A. The reaction is running in forward direction.
B. The reaction is running in reverse direction
C. The reaction is neither product nor reactant favored
1
answer
0
watching
586
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nelly StrackeLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232