1
answer
0
watching
2,196
views
18 Dec 2019
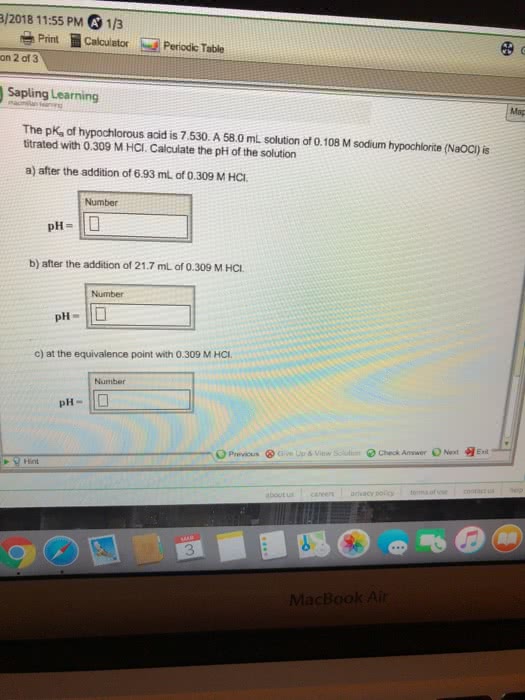
The pKa of hypochlorous acid is 7.530. A 56.0 mL solution of 0.145 M sodium hypochlorite (NaOCl) is titrated with 0.282 M HCl. Calculate the pH of the solution at: a) after the addition of 11.0 mL of 0.282 M HCl. b) after the addition of 30.3 mL of 0.282 M HCl. c) equivalence point of .282M HCl
The pKa of hypochlorous acid is 7.530. A 56.0 mL solution of 0.145 M sodium hypochlorite (NaOCl) is titrated with 0.282 M HCl. Calculate the pH of the solution at: a) after the addition of 11.0 mL of 0.282 M HCl. b) after the addition of 30.3 mL of 0.282 M HCl. c) equivalence point of .282M HCl
Nestor RutherfordLv2
31 Dec 2019