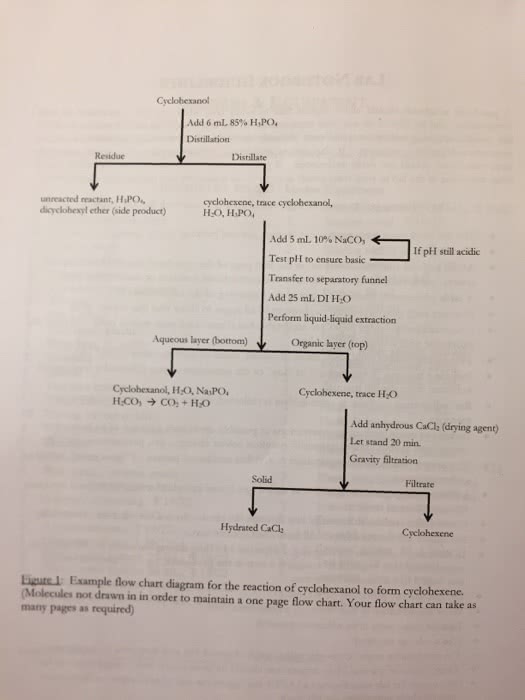
Procedure Reaction 1: Dissolving the Copper 1. Obtain a clean, dry, glass centrifuge tube. 2. Place a piece of copper wire in a weighing paper, determine the mass of the wire and place it in the centrifuge tube. The copper wire should weigh less than 0.0200 grams. 3. In a fume hood, add seven drops of concentrated nitric acid to the reaction tube so that the copper metal dissolves completely. Describe your observations in the lab report. (Caution, Concentrated nitric acid and nitrogen dioxide are very corrosive. Either will turn your skin yellow on contact. Do not leave any spills on the lab bench or in the fume hood.) 4. When the copper has dissolved, add seven drops of distilled water to the tube. Reaction 2: Preparation of Copper(II) Hydroxide 1. Add 15 drops of 3.0 M aqueous sodium hydroxide to the tube. Make sure that the reactants are well mixed. Shake the tube carefully or gently flick the bottom of the tube with your finger. Remember that the contents of the tube may still be corrosive. 2. Add a second 15 drops of NaOH(aq), mix well, and record your observations. If you have two layers at this point it means that you have not mixed the solution well enough. 3. Centrifuge the reaction mixture. 4. The liquid at the top of the centrifuged mixture is called the supernatant while the solid is called a precipitate. Before separating the supernatant from the precipitate it is necessary to ensure that all of the copper(II) hydroxide has been precipitated. The supernatant should be clear and colorless indicating the absence of any Cu+2 ions in the solution. It should also be basic due to an excess of OH- ions. Using a clean glass stirring rod, transfer a drop of the supernatant onto a piece of red litmus paper. If the litmus paper turns blue then the solution is basic and enough NaOH has been added. If the paper does not turn blue, add more NaOH, mix well, recentrifuge, and repeat the litmus paper test until the paper does turn blue. 5. An efficient separation of supernatants and precipitates is key to obtaining a good final yield of copper. The supernatant liquid can be separated from the precipitate by expelling the air from the bulb of a Pasteur pipet, inserting the tip of the pipet into the supernatant, then gently sucking the supernatant into the pipet. If you expel air or liquid into the precipitate with the pipet, you will stir up the precipitate and will have to repeat the centrifugation step. Remove as much liquid as possible and discard it in the waste container provided on the instructorâs cart. It is better to leave a small amount of supernatant liquid than to remove some of the copper(II) hydroxide precipitate. Reaction 3: Formation of Copper(II) Oxide 1. Set up a hot water bath by placing a beaker of water on a hotplate, placing an iron ring around the beaker, and heating the water to boiling. 2. Place the centrifuge tube containing the copper(II) hydroxide into the boiling water. Carefully hold the tube with a test tube clamp so that it doesnât get water into it. Record your observations. Reaction 4: Formation of Copper(II) Sulfate 1. Add 20 drops of 3.0 M H2SO4 to the solid in the centrifuge tube. Stir carefully to ensure that the copper(II) oxide dissolves completely. Complete dissolution of the mixture will require thorough mixing and possibly heating of the solution. 2. Obtain the mass of a small, clean, glass test tube as accurately as possible. 3. Transfer the liquid from the centrifuge tube into the test tube. Rinse the centrifuge carefully with 1.0 mL of distilled water and transfer the rinse into the test tube containing your sample. 4. Record your observations on the data sheet. Reaction 5: Formation of Copper Metal 1. Add a small quantity of zinc powder to the sample solution. Continue adding zinc in small quantities until the solution loses the blue copper(II) color. Any excess zinc added will need to be removed so donât add it too quickly or in large quantities. When the solution has turned colorless, add several drops of 3.0 M H2SO4 to the tube to dissolve any left over zinc. You can tell that the zinc has dissolved when addition of sulfuric acid does not generate bubbles. 2. Allow the copper metal to sink to the bottom of the tube and carefully remove the supernatant liquid using a Pasteur pipet. 3. Wash the red-brown copper metal in the tube with 1.0 mL of water. Allow the copper metal to settle to the bottom and remove the excess water. Repeat this rinsing process two more times. 4. Describe your observations on the data sheet. Drying the copper metal 1. After removing as much of the third rinse water as possible you are ready to dry the metal. This must be done carefully in a cool Bunsen Burner flame. If the tube is heated too quickly there is a risk of ejecting the contents of the tube as the water boils. Also, if the flame is too hot you may convert the copper metal back into black copper(II) oxide. The objective is to drive the water from the tube as steam. Make sure that as water condenses on the walls of the tube that you continue to heat until all of the water if gone from the tube. 2. Once all of the water is removed from the tube, cool the tube and its contents then determine the mass of copper by weighing the tube and subtracting the tube + copper weight from the weight of the empty tube (Reaction 4 step 2). If the mass of copper is higher than the original mass of the copper wire it either contains water or zinc or has been converted to copper(II) oxide. Excess water can be removed by reheating the tube and reweighing to constant mass. Excess zinc requires addition of sulfuric acid followed by re-rinsing with water and re-drying. Chemistry 1215 Experiment 9 Lab Report Name ______________________________ Data Sheet Mass of copper wire _______________ Mass of clean, dry test tube _______________ Mass of test tube plus copper _______________ Mass of final copper sample _______________ Percent recovery of copper. Show all calculations. Observations 1. Describe your observations for Reaction 1 including colors, gases formed, etc. 2. Describe your observations for Reaction 2 including colors, gases formed, etc. 3. Describe your observations for Reaction 3 including colors, gases formed, etc. Estimate the temperature of the decomposition of Copper(II) hydroxide. 4. Describe your observations for Reaction 4 including colors, gases formed, etc. 5. Describe your observations for Reaction 5 including colors, gases formed, etc. Write a brief discussion of your results including a statement of the final percent recovery of copper and a discussion of reasons why the recovery differs from 100%. Chemistry 1215, Experiment #9; Copper and its compounds, Pre-lab Name ____________________________________ 1. Write a balanced chemical equation including phase labels for the reaction between aqueous copper (II) nitrate and aqueous sodium hydroxide. 2. Nitrogen monoxide (NO) and nitrogen dioxide (NO2) are toxic, corrosive gases that significantly lower blood pressure when inhaled. How are these gases produced in todayâs experiment? What should you do to protect yourself against their toxicity? 3. Iron reacts with oxygen from the atmosphere to produce iron (III) oxide, also known as rust (Fe2O3). What chemical species is oxidized in this reaction? What is the reducing agent? Jaffrey Zagnut couldnât find any nitric acid so he tried to dissolve his copper sample in hydrochloric acid instead. Unfortunately his copper wouldnât dissolve in HCl. Why will copper dissolve in nitric acid but not in hydrochloric acid (after all, HCl is a stronger acid than HNO3). Chemistry 1215, Experiment #9; Copper and its compounds, Post-lab Name ____________________________________ 1. Copper (II) hydroxide is converted into copper (II) oxide by heating the test tube containing Cu(OH)2 in a hot water bath. Is it necessary to use distilled water in this water bath? Why or why not? 2. Copper metal doesnât ârustâ in the presence of oxygen at room temperature. However, it will react with O2 at elevated temperatures. Write a balanced chemical equation describing the formation of copper (II) oxide when copper metal is heated in air. 3. When zinc is dissolved in sulfuric acid a gas is produced. What is the chemical identity of this gas? How is it produced? 4. Jaffrey Zagnut started with a 0.032 g sample of copper which he took through the series of reactions described in this experiment. At the end of the experiment he obtained 0.038 g of a black product. What was his percent yield? What is the most likely source of the error in his experiment? (Hint: consider question 2 above)
Procedure Reaction 1: Dissolving the Copper 1. Obtain a clean, dry, glass centrifuge tube. 2. Place a piece of copper wire in a weighing paper, determine the mass of the wire and place it in the centrifuge tube. The copper wire should weigh less than 0.0200 grams. 3. In a fume hood, add seven drops of concentrated nitric acid to the reaction tube so that the copper metal dissolves completely. Describe your observations in the lab report. (Caution, Concentrated nitric acid and nitrogen dioxide are very corrosive. Either will turn your skin yellow on contact. Do not leave any spills on the lab bench or in the fume hood.) 4. When the copper has dissolved, add seven drops of distilled water to the tube. Reaction 2: Preparation of Copper(II) Hydroxide 1. Add 15 drops of 3.0 M aqueous sodium hydroxide to the tube. Make sure that the reactants are well mixed. Shake the tube carefully or gently flick the bottom of the tube with your finger. Remember that the contents of the tube may still be corrosive. 2. Add a second 15 drops of NaOH(aq), mix well, and record your observations. If you have two layers at this point it means that you have not mixed the solution well enough. 3. Centrifuge the reaction mixture. 4. The liquid at the top of the centrifuged mixture is called the supernatant while the solid is called a precipitate. Before separating the supernatant from the precipitate it is necessary to ensure that all of the copper(II) hydroxide has been precipitated. The supernatant should be clear and colorless indicating the absence of any Cu+2 ions in the solution. It should also be basic due to an excess of OH- ions. Using a clean glass stirring rod, transfer a drop of the supernatant onto a piece of red litmus paper. If the litmus paper turns blue then the solution is basic and enough NaOH has been added. If the paper does not turn blue, add more NaOH, mix well, recentrifuge, and repeat the litmus paper test until the paper does turn blue. 5. An efficient separation of supernatants and precipitates is key to obtaining a good final yield of copper. The supernatant liquid can be separated from the precipitate by expelling the air from the bulb of a Pasteur pipet, inserting the tip of the pipet into the supernatant, then gently sucking the supernatant into the pipet. If you expel air or liquid into the precipitate with the pipet, you will stir up the precipitate and will have to repeat the centrifugation step. Remove as much liquid as possible and discard it in the waste container provided on the instructorâs cart. It is better to leave a small amount of supernatant liquid than to remove some of the copper(II) hydroxide precipitate. Reaction 3: Formation of Copper(II) Oxide 1. Set up a hot water bath by placing a beaker of water on a hotplate, placing an iron ring around the beaker, and heating the water to boiling. 2. Place the centrifuge tube containing the copper(II) hydroxide into the boiling water. Carefully hold the tube with a test tube clamp so that it doesnât get water into it. Record your observations. Reaction 4: Formation of Copper(II) Sulfate 1. Add 20 drops of 3.0 M H2SO4 to the solid in the centrifuge tube. Stir carefully to ensure that the copper(II) oxide dissolves completely. Complete dissolution of the mixture will require thorough mixing and possibly heating of the solution. 2. Obtain the mass of a small, clean, glass test tube as accurately as possible. 3. Transfer the liquid from the centrifuge tube into the test tube. Rinse the centrifuge carefully with 1.0 mL of distilled water and transfer the rinse into the test tube containing your sample. 4. Record your observations on the data sheet. Reaction 5: Formation of Copper Metal 1. Add a small quantity of zinc powder to the sample solution. Continue adding zinc in small quantities until the solution loses the blue copper(II) color. Any excess zinc added will need to be removed so donât add it too quickly or in large quantities. When the solution has turned colorless, add several drops of 3.0 M H2SO4 to the tube to dissolve any left over zinc. You can tell that the zinc has dissolved when addition of sulfuric acid does not generate bubbles. 2. Allow the copper metal to sink to the bottom of the tube and carefully remove the supernatant liquid using a Pasteur pipet. 3. Wash the red-brown copper metal in the tube with 1.0 mL of water. Allow the copper metal to settle to the bottom and remove the excess water. Repeat this rinsing process two more times. 4. Describe your observations on the data sheet. Drying the copper metal 1. After removing as much of the third rinse water as possible you are ready to dry the metal. This must be done carefully in a cool Bunsen Burner flame. If the tube is heated too quickly there is a risk of ejecting the contents of the tube as the water boils. Also, if the flame is too hot you may convert the copper metal back into black copper(II) oxide. The objective is to drive the water from the tube as steam. Make sure that as water condenses on the walls of the tube that you continue to heat until all of the water if gone from the tube. 2. Once all of the water is removed from the tube, cool the tube and its contents then determine the mass of copper by weighing the tube and subtracting the tube + copper weight from the weight of the empty tube (Reaction 4 step 2). If the mass of copper is higher than the original mass of the copper wire it either contains water or zinc or has been converted to copper(II) oxide. Excess water can be removed by reheating the tube and reweighing to constant mass. Excess zinc requires addition of sulfuric acid followed by re-rinsing with water and re-drying. Chemistry 1215 Experiment 9 Lab Report Name ______________________________ Data Sheet Mass of copper wire _______________ Mass of clean, dry test tube _______________ Mass of test tube plus copper _______________ Mass of final copper sample _______________ Percent recovery of copper. Show all calculations. Observations 1. Describe your observations for Reaction 1 including colors, gases formed, etc. 2. Describe your observations for Reaction 2 including colors, gases formed, etc. 3. Describe your observations for Reaction 3 including colors, gases formed, etc. Estimate the temperature of the decomposition of Copper(II) hydroxide. 4. Describe your observations for Reaction 4 including colors, gases formed, etc. 5. Describe your observations for Reaction 5 including colors, gases formed, etc. Write a brief discussion of your results including a statement of the final percent recovery of copper and a discussion of reasons why the recovery differs from 100%. Chemistry 1215, Experiment #9; Copper and its compounds, Pre-lab Name ____________________________________ 1. Write a balanced chemical equation including phase labels for the reaction between aqueous copper (II) nitrate and aqueous sodium hydroxide. 2. Nitrogen monoxide (NO) and nitrogen dioxide (NO2) are toxic, corrosive gases that significantly lower blood pressure when inhaled. How are these gases produced in todayâs experiment? What should you do to protect yourself against their toxicity? 3. Iron reacts with oxygen from the atmosphere to produce iron (III) oxide, also known as rust (Fe2O3). What chemical species is oxidized in this reaction? What is the reducing agent? Jaffrey Zagnut couldnât find any nitric acid so he tried to dissolve his copper sample in hydrochloric acid instead. Unfortunately his copper wouldnât dissolve in HCl. Why will copper dissolve in nitric acid but not in hydrochloric acid (after all, HCl is a stronger acid than HNO3). Chemistry 1215, Experiment #9; Copper and its compounds, Post-lab Name ____________________________________ 1. Copper (II) hydroxide is converted into copper (II) oxide by heating the test tube containing Cu(OH)2 in a hot water bath. Is it necessary to use distilled water in this water bath? Why or why not? 2. Copper metal doesnât ârustâ in the presence of oxygen at room temperature. However, it will react with O2 at elevated temperatures. Write a balanced chemical equation describing the formation of copper (II) oxide when copper metal is heated in air. 3. When zinc is dissolved in sulfuric acid a gas is produced. What is the chemical identity of this gas? How is it produced? 4. Jaffrey Zagnut started with a 0.032 g sample of copper which he took through the series of reactions described in this experiment. At the end of the experiment he obtained 0.038 g of a black product. What was his percent yield? What is the most likely source of the error in his experiment? (Hint: consider question 2 above)