0
answers
0
watching
130
views
18 Dec 2019
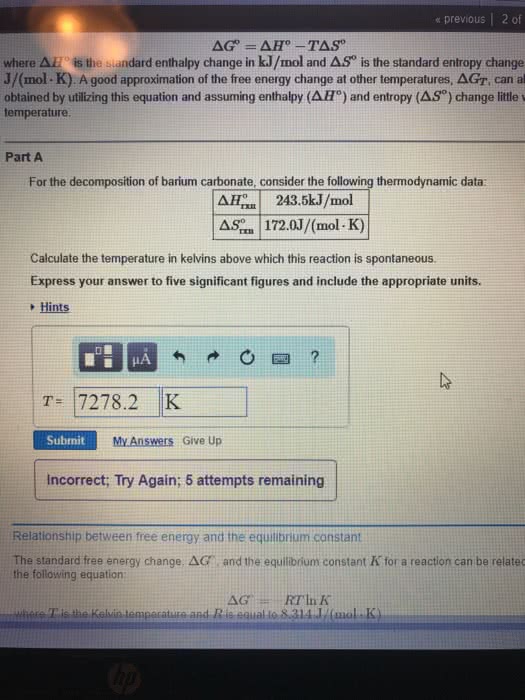
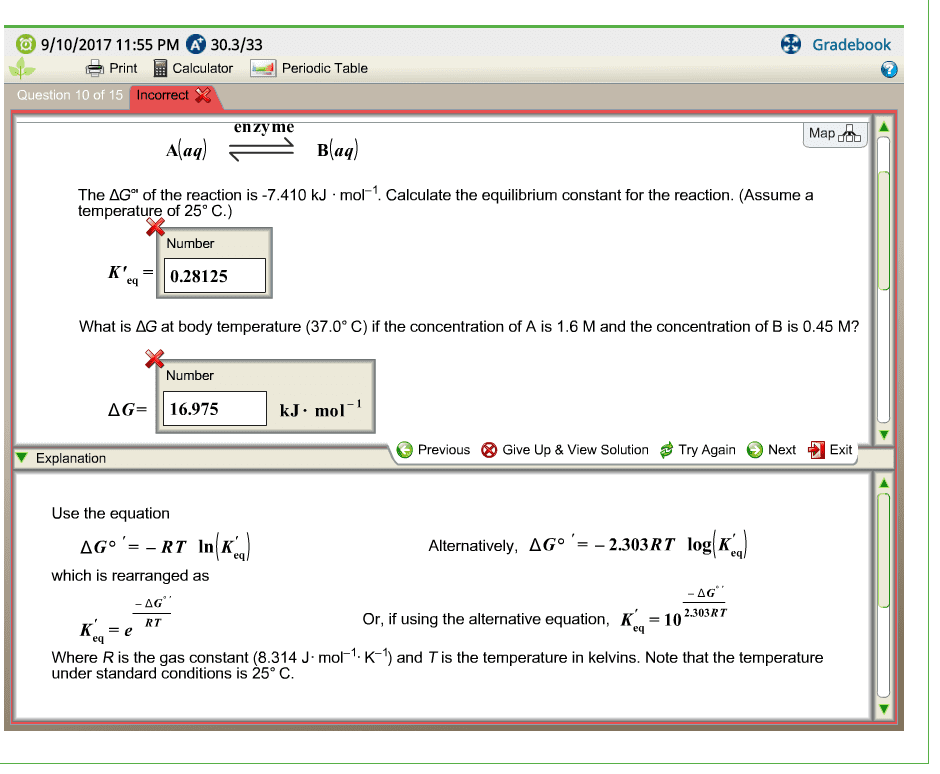
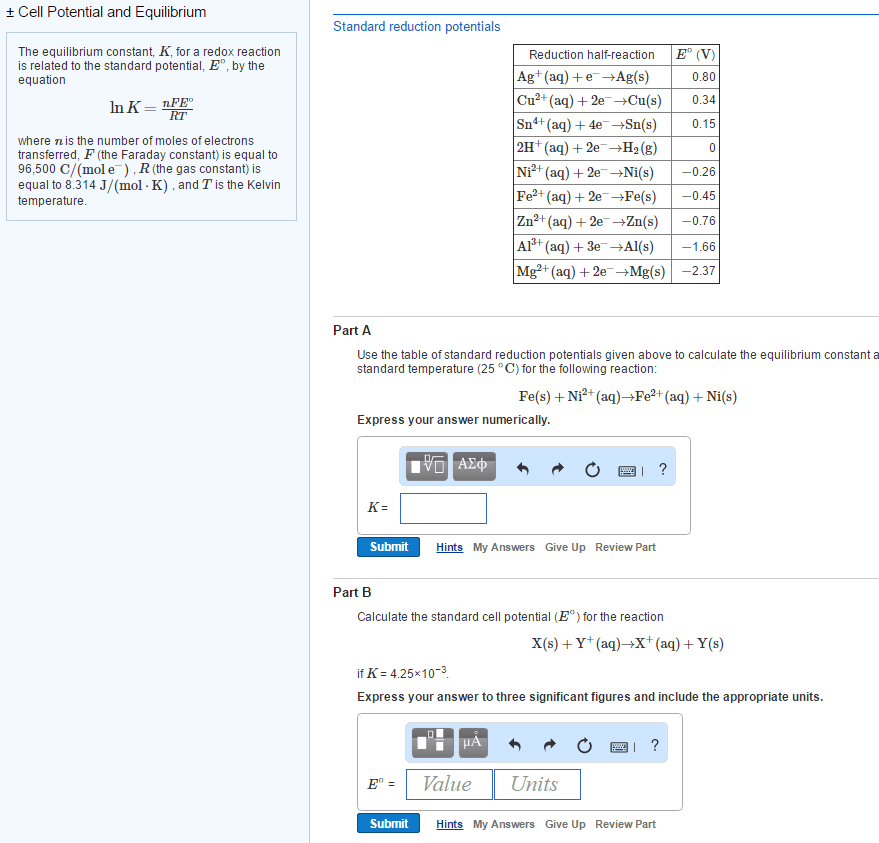
Relationship between free energy and the equilibrium constantThe standard free energy change Delta G^\circ and the equilibrium constant K for a reaction can be related by the following equation:Delta G^\circ=-RT\ln Kwhere T is the Kelvin temperature and R is equal to \rm 8.314 J/(mol\cdot K).Part BCalculate the equilibrium constant for the reaction forming nitric oxide at room temperature, 25 ^{\circ}C.
Relationship between free energy and the equilibrium constantThe standard free energy change Delta G^\circ and the equilibrium constant K for a reaction can be related by the following equation:Delta G^\circ=-RT\ln Kwhere T is the Kelvin temperature and R is equal to \rm 8.314 J/(mol\cdot K).Part BCalculate the equilibrium constant for the reaction forming nitric oxide at room temperature, 25 ^{\circ}C.
0
answers
0
watching
130
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232