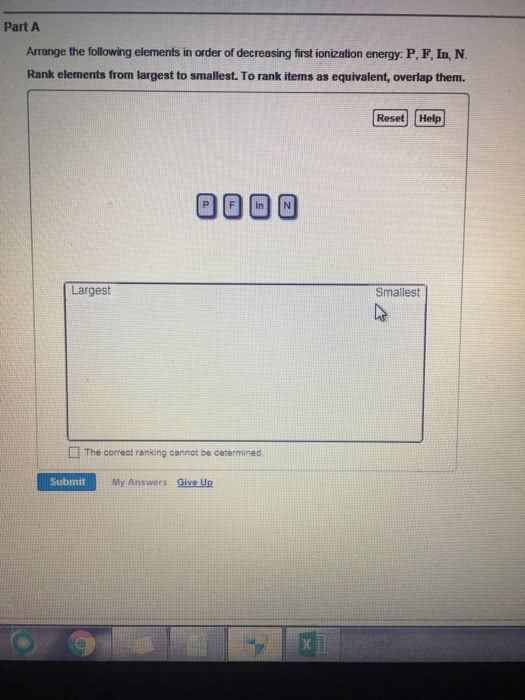
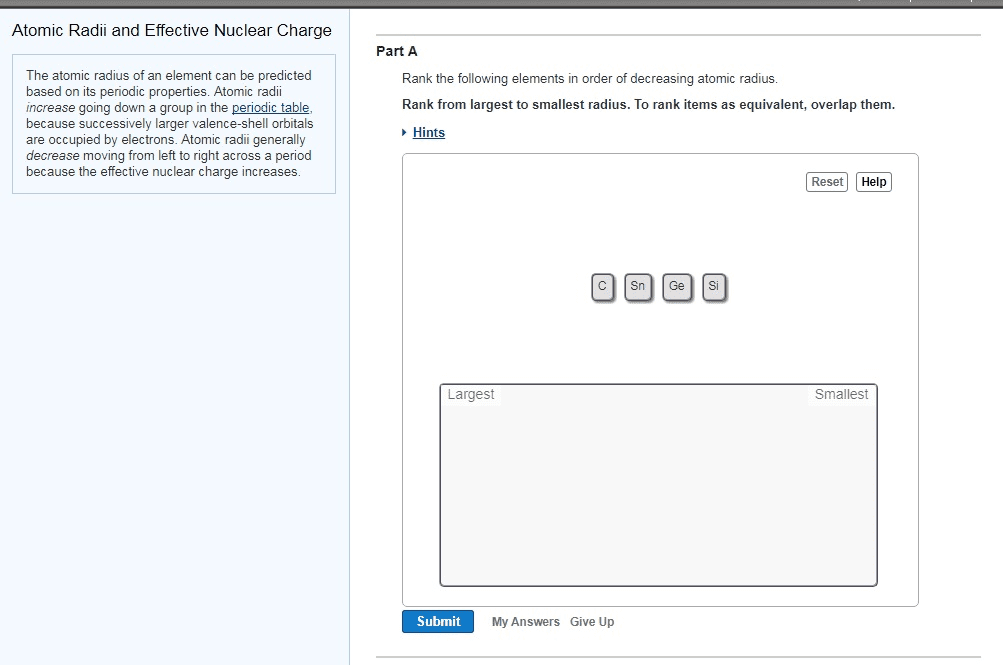
1
answer
0
watching
427
views
18 Dec 2019
A)Each element in the periodic table has a distinctive atomic radius.Place the following elements in order of decreasing atomic size: selenium, chlorine, fluorine, rubidium, calcium, and sulfur.Rank from largest to smallest. To rank items as equivalent, overlap them.B)Arrange the following elements in order of decreasing atomic radius: Ba, Sn, S, Tl, Se.Rank elements from largest to smallest. To rank items as equivalent, overlap them.
A)Each element in the periodic table has a distinctive atomic radius.Place the following elements in order of decreasing atomic size: selenium, chlorine, fluorine, rubidium, calcium, and sulfur.Rank from largest to smallest. To rank items as equivalent, overlap them.B)Arrange the following elements in order of decreasing atomic radius: Ba, Sn, S, Tl, Se.Rank elements from largest to smallest. To rank items as equivalent, overlap them.
Patrina SchowalterLv2
31 Dec 2019