1
answer
0
watching
82
views
18 Dec 2019
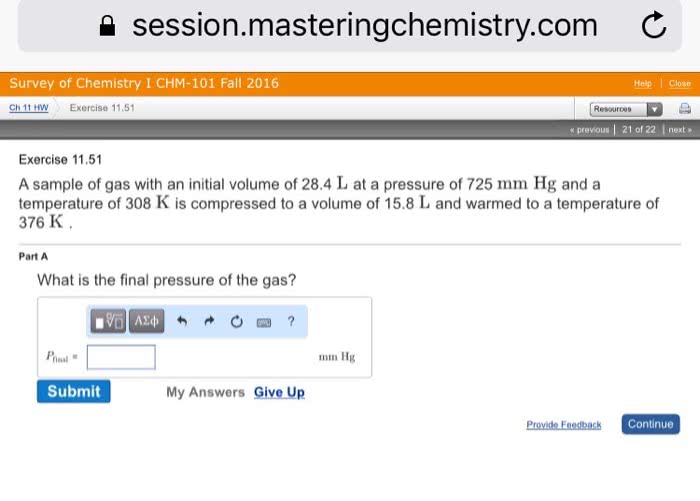
A sample of gas with an initial volume of 34.5 L at a pressure of 765 mm Hg and a temperature of 325 K is compressed to a volume of 16.0 L and warmed to a temperature of 400 K. What is the final pressure of the gas?
A sample of gas with an initial volume of 34.5 L at a pressure of 765 mm Hg and a temperature of 325 K is compressed to a volume of 16.0 L and warmed to a temperature of 400 K. What is the final pressure of the gas?
Reid WolffLv2
31 Dec 2019