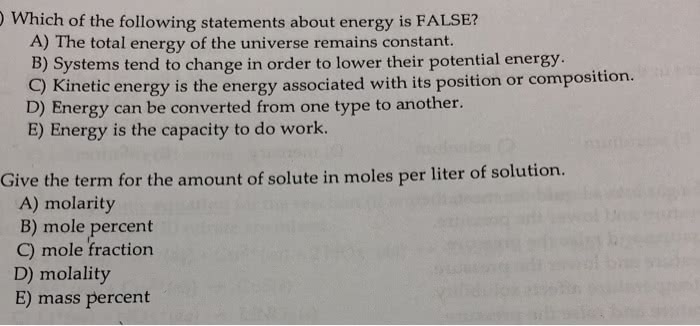1
answer
0
watching
105
views
18 Dec 2019
Which of the following statements about the first law ofthermodynamics and energy is false?
A.) Kinetic energy can be converted to potential energy.
B.) Kinetic energy = 1/2 mv.
C.) All the energy in the universe is conserved.
D.) A system can never decrease its energy.
E.)Potential energy is the energy of position or composition.
Which of the following statements about the first law ofthermodynamics and energy is false?
A.) Kinetic energy can be converted to potential energy.
B.) Kinetic energy = 1/2 mv.
C.) All the energy in the universe is conserved.
D.) A system can never decrease its energy.
E.)Potential energy is the energy of position or composition.
Nestor RutherfordLv2
31 Dec 2019