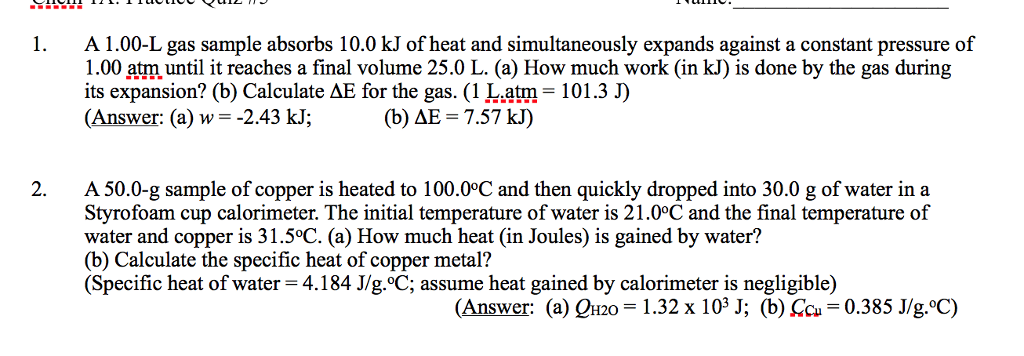1
answer
0
watching
104
views
6 Apr 2020
You find a penny (assume to be pure copper with a specific heat of 0.385 J/g°C) in the snow which has a temperature of -10.0°C. How much heat, in Joules, is absorbed by the penny as it warms to body temperature, 35.0°C? The mass of the penny is 3.10 g. Report answer to 3 significant figures and use + or - to indicate the direction of energy flow. Assume the unit to be Joules.
You find a penny (assume to be pure copper with a specific heat of 0.385 J/g°C) in the snow which has a temperature of -10.0°C. How much heat, in Joules, is absorbed by the penny as it warms to body temperature, 35.0°C? The mass of the penny is 3.10 g. Report answer to 3 significant figures and use + or - to indicate the direction of energy flow. Assume the unit to be Joules.
1
answer
0
watching
104
views
For unlimited access to Homework Help, a Homework+ subscription is required.
2 Jun 2021
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232