1
answer
0
watching
528
views
28 Apr 2020
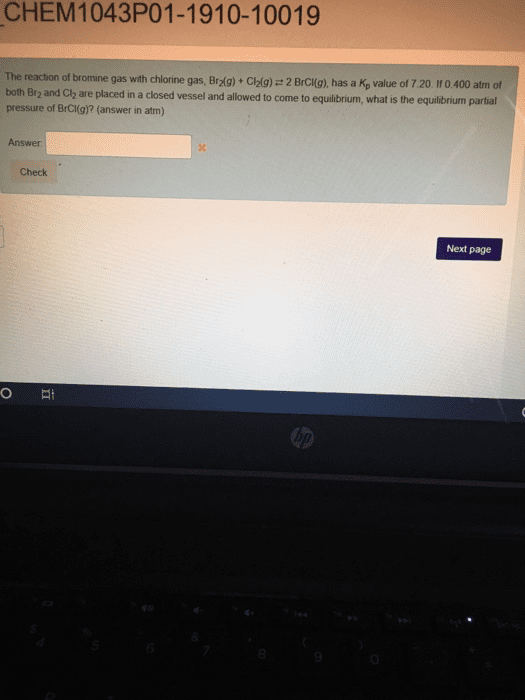
The reaction of bromine gas with chlorine gas, shown here, has a Kc value of 7.20 at 200ºC. If a closed vessel was charged with the two reactants, each at an initial concentration of 0.200 M, but with no initial concentration of BrCl, what would be the equilibrium concentration of BrCl(g)?
Br2(g) + Cl2(g)= 2BrCl(g) K = 7.20
Initial Concentrations 0.20 0.20 0
The reaction of bromine gas with chlorine gas, shown here, has a Kc value of 7.20 at 200ºC. If a closed vessel was charged with the two reactants, each at an initial concentration of 0.200 M, but with no initial concentration of BrCl, what would be the equilibrium concentration of BrCl(g)?
Br2(g) + Cl2(g)= 2BrCl(g) K = 7.20
Initial Concentrations 0.20 0.20 0
Robert KubaraLv10
14 Feb 2021