1
answer
0
watching
244
views
7 May 2020
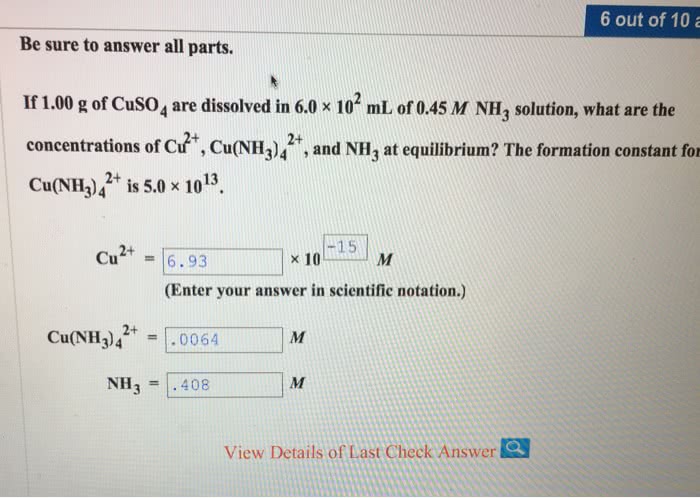
If 2.50 g of CuSO4 are dissolved in 6.0 × 102 mL of 0.45 M NH3 solution, what are the concentrations of Cu2+, Cu(NH3)42+, and NH3 at equilibrium? The formation constant for Cu(NH3)42+ is 5.0 × 1013.
If 2.50 g of CuSO4 are dissolved in 6.0 × 102 mL of 0.45 M NH3 solution, what are the concentrations of Cu2+, Cu(NH3)42+, and NH3 at equilibrium? The formation constant for Cu(NH3)42+ is 5.0 × 1013.
Read by 4 people
2 Jun 2021