1
answer
0
watching
588
views
28 Mar 2020
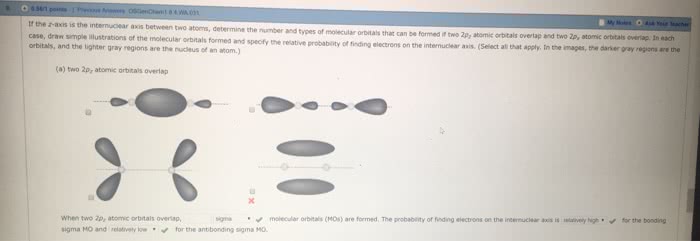
(a) What is the probability of finding an electron on the internuclear axis if the electron occupies a π molecular orbital? (b) For a homonuclear diatomic molecule, what similarities and differences are there between the π2p MO made from the 2px atomic orbitals and the π2p MO made from the 2py atomic orbitals? (c) How do the π*2p MOs formed from the 2px and 2py atomic orbitals differ from the π2p MOs in terms of energies and electron distributions?
(a) What is the probability of finding an electron on the internuclear axis if the electron occupies a π molecular orbital? (b) For a homonuclear diatomic molecule, what similarities and differences are there between the π2p MO made from the 2px atomic orbitals and the π2p MO made from the 2py atomic orbitals? (c) How do the π*2p MOs formed from the 2px and 2py atomic orbitals differ from the π2p MOs in terms of energies and electron distributions?
Deanna HettingerLv2
20 May 2020