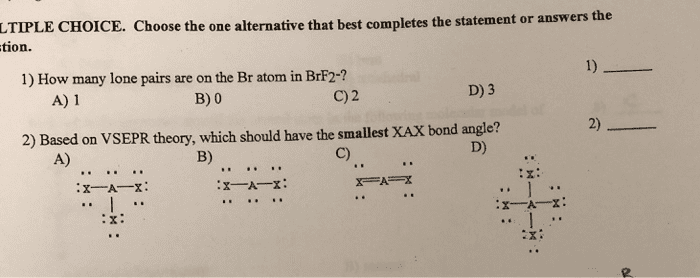The azide ion, N3–, is linear with two N–N bonds of equal length, 1.16 Å. (a) Draw a Lewis structure for the azide ion. (b) With reference to Table 8.5, is the observed N–N bond length consistent with your Lewis structure? (c) What hybridization scheme would you expect at each of the nitrogen atoms in N3–? (d) Show which hybridized and unhybridized orbitals are involved in the formation of σ and π bonds in N3–. (e) It is often observed that σ bonds that involve an sp hybrid orbital are shorter than those that involve only sp2 or sp3 hybrid orbitals. Can you propose a reason for this? Is this observation applicable to the observed bond lengths in N3–?
The azide ion, N3–, is linear with two N–N bonds of equal length, 1.16 Å. (a) Draw a Lewis structure for the azide ion. (b) With reference to Table 8.5, is the observed N–N bond length consistent with your Lewis structure? (c) What hybridization scheme would you expect at each of the nitrogen atoms in N3–? (d) Show which hybridized and unhybridized orbitals are involved in the formation of σ and π bonds in N3–. (e) It is often observed that σ bonds that involve an sp hybrid orbital are shorter than those that involve only sp2 or sp3 hybrid orbitals. Can you propose a reason for this? Is this observation applicable to the observed bond lengths in N3–?
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Help!
1)Match each molecule with the hybridization of the chlorine atom.
| A | sp2 |
| B | sp3d |
| C | sp3 |
| D | sp |
| E | sp3d2 |
Match the letter
ClO4-
ClO2
ClO3-
ClO2-
2)Match each electron group arrangement with the corresponding central-atom orbital hybridization.
| A | sp3 |
| B | sp2 |
| C | sp3d2 |
| D | sp3d |
| E | sp |
Match the letter
tetrahedral
octahedral
trigonal planar
trigonal bipyramidal
linear
3)Like several other bonds, carbon-oxygen bonds have lengths and strengths that depend on the bond order. Draw Lewis structures for the following species, and arrange them in order of increasing carbon-oxygen bond length. (Indicate the order with the numbers from the drop-down list, starting with 1 for the compound with the smallest bond length to 5 for the compound with the largest bond length.)
CO32-
HCO3-(H attached to O)
H2CO
CO
CH4O
4)Which have a molecular dipole moment?
Yes, No SF6
Yes No CS2
Yes No PF3
Yes No SF4
Yes No F2