1
answer
2
watching
491
views
6 Apr 2020
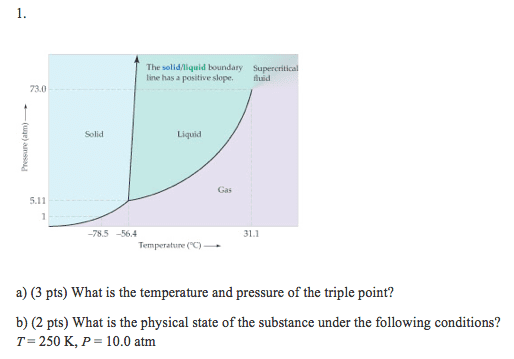
The phase diagram of a hypothetical substance is

(a) Estimate the normal boiling point and freezing point of the substance.
(b) What is the physical state of the substance under the following conditions? (i) T = 150 K, P = 0.2 atm; (ii) T = 100 K, P = 0.8 atm; (iii) T = 300 K, P = 1.0 atm.
(c) What is the triple point of the substance?
The phase diagram of a hypothetical substance is
(a) Estimate the normal boiling point and freezing point of the substance.
(b) What is the physical state of the substance under the following conditions? (i) T = 150 K, P = 0.2 atm; (ii) T = 100 K, P = 0.8 atm; (iii) T = 300 K, P = 1.0 atm.
(c) What is the triple point of the substance?
Sixta KovacekLv2
13 May 2020