1
answer
0
watching
306
views
18 Mar 2020
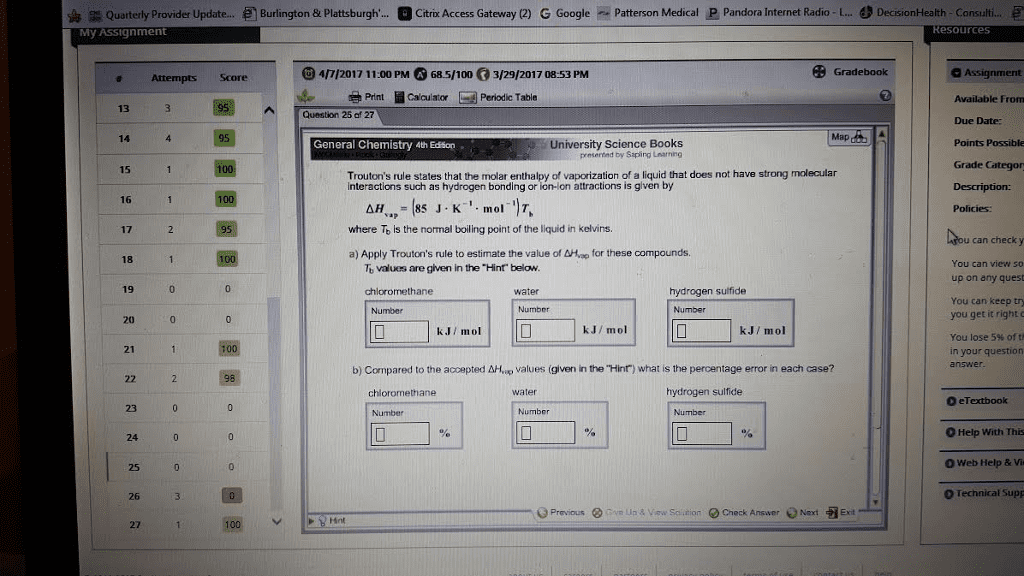
Trouton’s rule states that for many liquids at their normal boiling points, the standard molar entropy of vaporization is about 88 J/mol-K. (a) Estimate the normal boiling point of bromine, Br2, by determining ∆H°vap for Br2 using data from Appendix C. Assume that ∆H°vap remains constant with temperature and that Trouton’s rule holds. (b) Look up the normal boiling point of Br2 in a chemistry handbook or at the WebElements Web site (www.webelements. com) and compare it to your calculation. What are the possible sources of error, or incorrect assumptions, in the calculation?
Trouton’s rule states that for many liquids at their normal boiling points, the standard molar entropy of vaporization is about 88 J/mol-K. (a) Estimate the normal boiling point of bromine, Br2, by determining ∆H°vap for Br2 using data from Appendix C. Assume that ∆H°vap remains constant with temperature and that Trouton’s rule holds. (b) Look up the normal boiling point of Br2 in a chemistry handbook or at the WebElements Web site (www.webelements. com) and compare it to your calculation. What are the possible sources of error, or incorrect assumptions, in the calculation?
Tod ThielLv2
14 May 2020