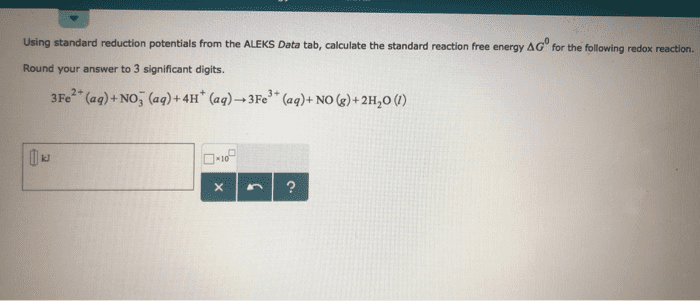
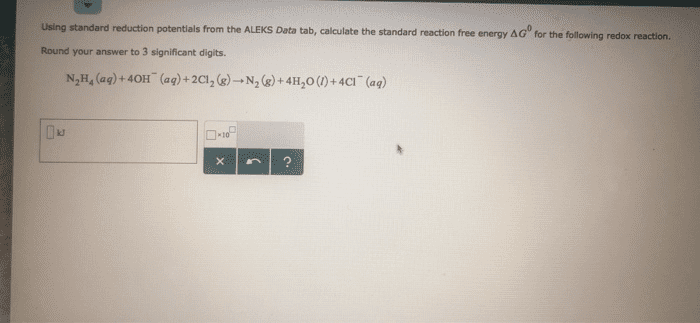
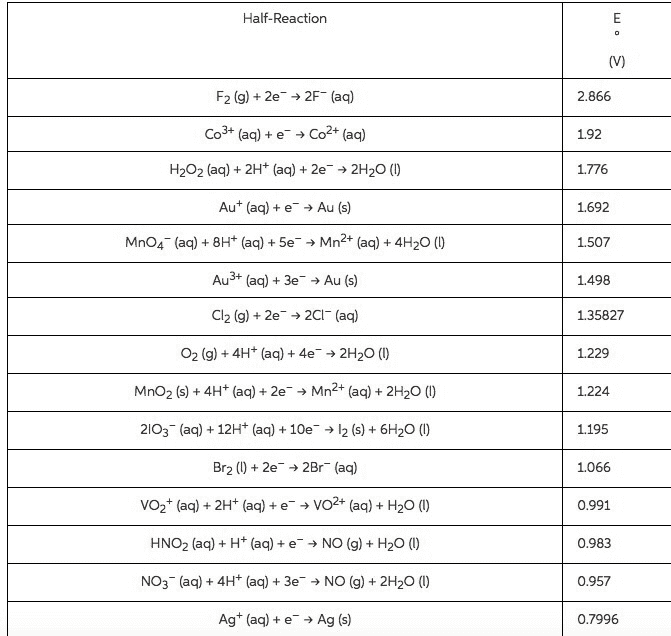
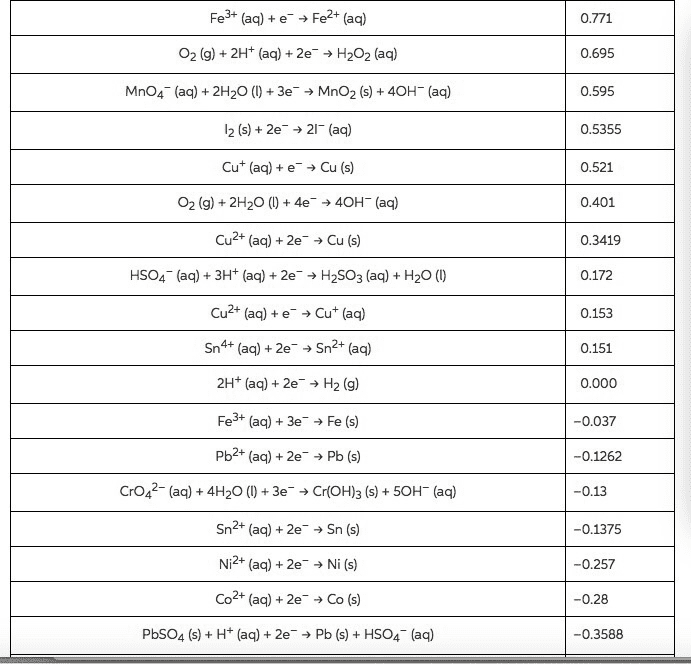
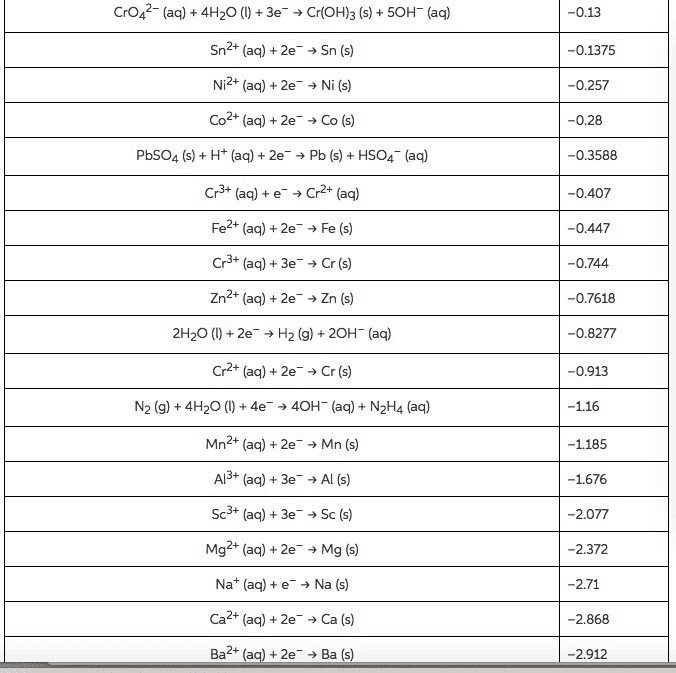
Using data in Appendix E, calculate the standard emf for each of the following reactions:
(a) H2(g) + F2(g)→2H+(aq) + 2F-(g)
(b) Cu2+(aq) + Ca(s)¡ →Cu(s) + Ca2+(aq)
(c) 3 Fe2+(aq)→Fe(aq) + 2Fe3+(aq)
(d) 2 ClO3-(aq) + 10 Br-(aq) + 12 H+(aq)→Cl2(g) + 5 Br2(l) + 6 H2O(g)
One Class Solution:-
(a)
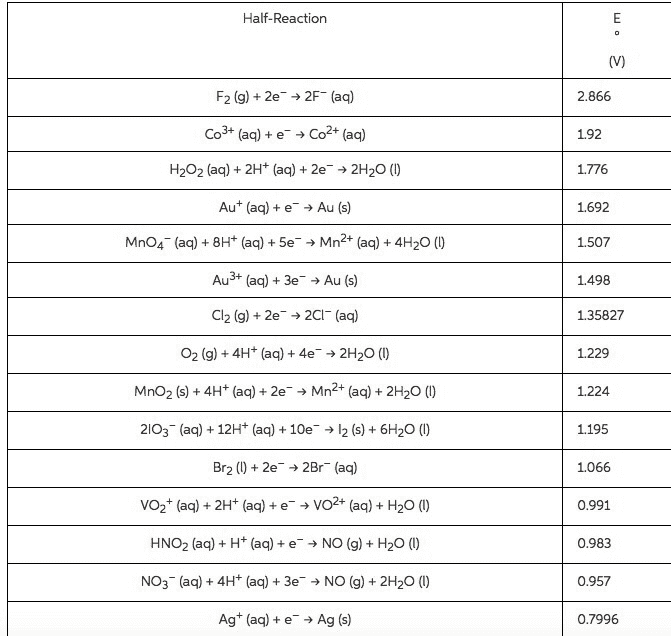
H2(g) →2H+(aq) +2e- , Eo = +0.0 V
F2(g) +2e-→ + 2F-(g), Eo = +2.87 V
Eo=2.87V
b)
Cu2+(aq) + 2e- →Cu(s) red. Eo=+0.337 V
Ca(s)→ 2e- + Ca2+(aq) oxid. Eo=-2.87 V
Eo=0.337- (-2.87)=3.207 V
c)
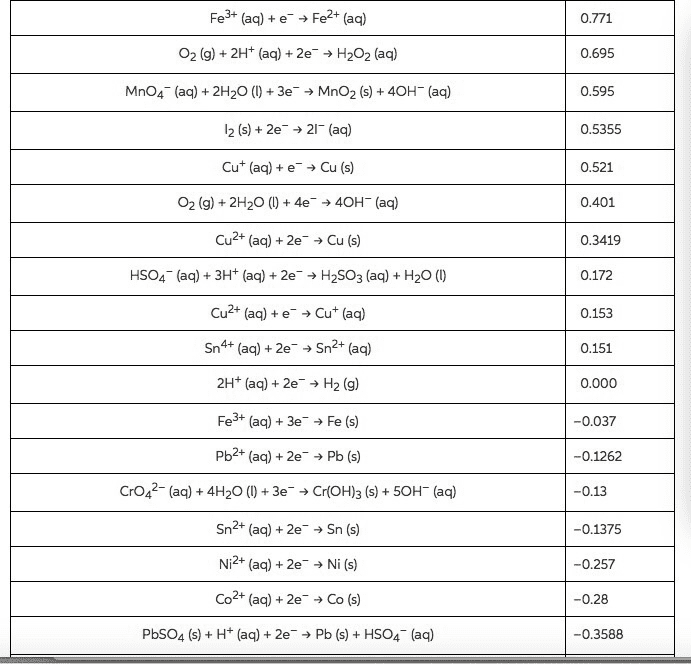
Fe2+(aq) +2e-→Fe(s) Eo=-0.440V
Fe2+(aq)→ e- + 2Fe3+(aq) 0.771 V
Eo= -0.440-0.771= -1.211V
(d) 2ClO3-(aq) + 10Br-(aq) + 12 H+(aq)→ Cl2(g) + 5Br2(l) + 6 H2O (l)
e)
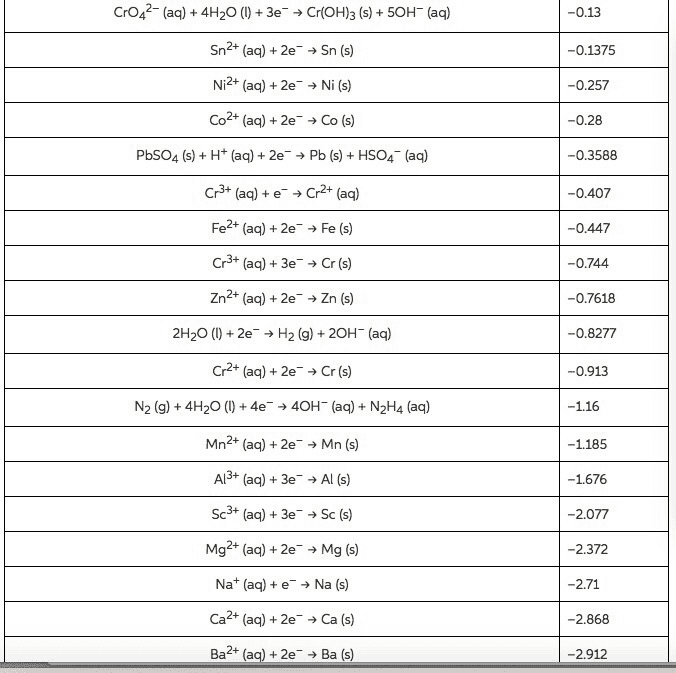
2ClO3-(aq) +10e- + 12 H+(aq)→ Cl2(g) + 6 H2O (l), Eo=+1.47 V
10 Br- (aq) → 5Br2(l) +2e-, Eo=1.0655 V
Eo=1.47-1.0655=0.4045V
Using data in Appendix E, calculate the standard emf for each of the following reactions:
(a) H2(g) + F2(g)→2H+(aq) + 2F-(g)
(b) Cu2+(aq) + Ca(s)¡ →Cu(s) + Ca2+(aq)
(c) 3 Fe2+(aq)→Fe(aq) + 2Fe3+(aq)
(d) 2 ClO3-(aq) + 10 Br-(aq) + 12 H+(aq)→Cl2(g) + 5 Br2(l) + 6 H2O(g)
One Class Solution:-
(a)
H2(g) →2H+(aq) +2e- , Eo = +0.0 V
F2(g) +2e-→ + 2F-(g), Eo = +2.87 V
Eo=2.87V
b)
Cu2+(aq) + 2e- →Cu(s) red. Eo=+0.337 V
Ca(s)→ 2e- + Ca2+(aq) oxid. Eo=-2.87 V
Eo=0.337- (-2.87)=3.207 V
c)
Fe2+(aq) +2e-→Fe(s) Eo=-0.440V
Fe2+(aq)→ e- + 2Fe3+(aq) 0.771 V
Eo= -0.440-0.771= -1.211V
(d) 2ClO3-(aq) + 10Br-(aq) + 12 H+(aq)→ Cl2(g) + 5Br2(l) + 6 H2O (l)
e)
2ClO3-(aq) +10e- + 12 H+(aq)→ Cl2(g) + 6 H2O (l), Eo=+1.47 V
10 Br- (aq) → 5Br2(l) +2e-, Eo=1.0655 V
Eo=1.47-1.0655=0.4045V
For unlimited access to Homework Help, a Homework+ subscription is required.