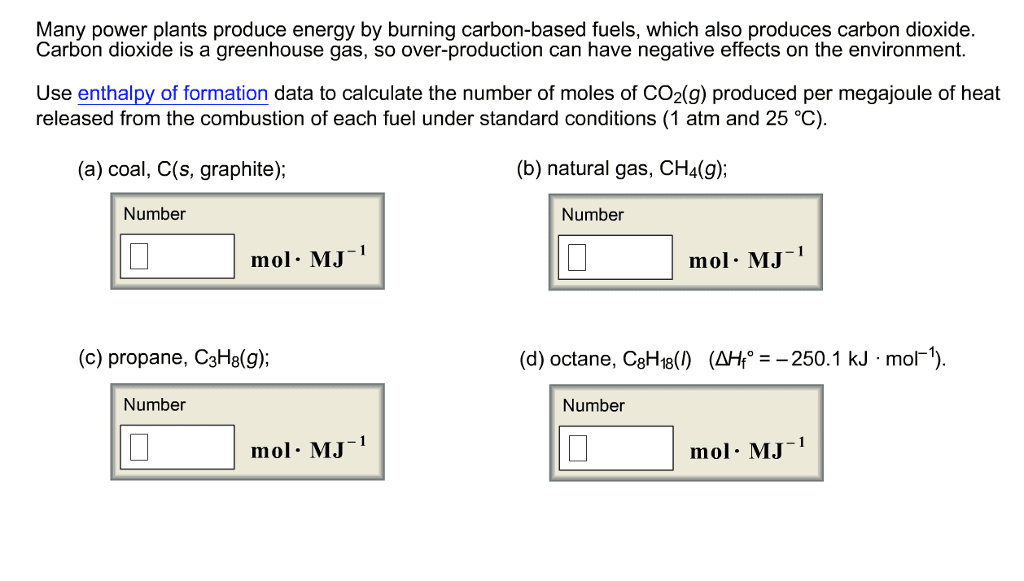
1
answer
0
watching
347
views
greyhound475Lv1
18 Apr 2020
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction:
4 

 + 2
+ 2 
Assume that all the products of the combustion of octane are in their gas phases. Use data from Exercise 21.50, Appendix C, and the inside covers of the text. The standard enthalpy of formation of octane is –250.1 kJ/mol.
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction:
4
+ 2
Assume that all the products of the combustion of octane are in their gas phases. Use data from Exercise 21.50, Appendix C, and the inside covers of the text. The standard enthalpy of formation of octane is –250.1 kJ/mol.
Casey DurganLv2
22 May 2020