1
answer
0
watching
77
views
goldpug979Lv1
6 Oct 2020
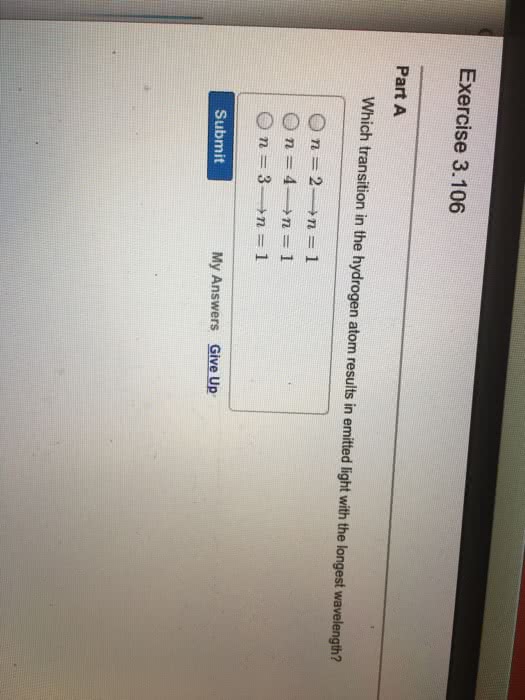
what is the wavelength of the light emitted when each of the following transitions occurs for a hydrogen atom? What type of EM radiation is emitted in each transition?
n=4 to n=3
n=5 to n=4
n=5 to n=3
what is the wavelength of the light emitted when each of the following transitions occurs for a hydrogen atom? What type of EM radiation is emitted in each transition?
n=4 to n=3
n=5 to n=4
n=5 to n=3
Lulama KlaasLv10
28 Dec 2020