1
answer
0
watching
490
views
6 Oct 2020
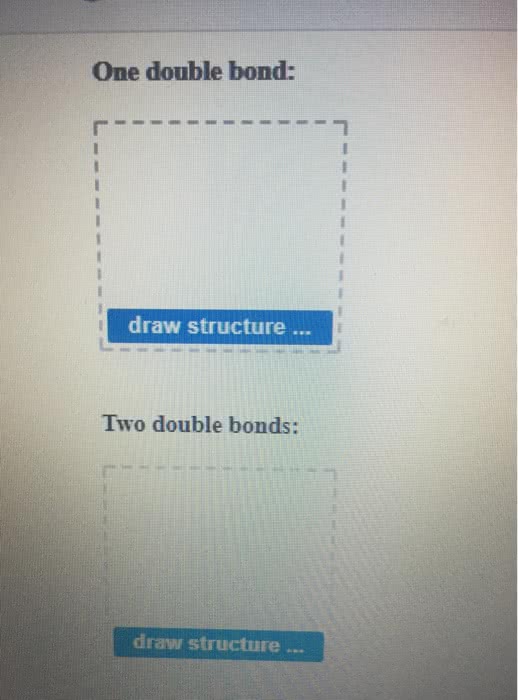
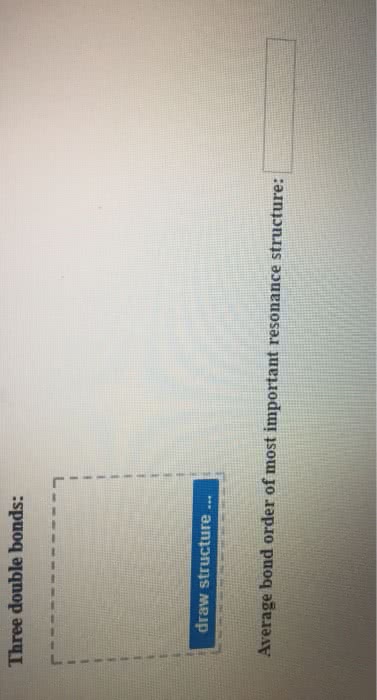
Several Lewis structures can be written for perbromate ion, BrO4-, the central Br with all single Br-O bonds, or with one, two, or three Br=O double bonds. Draw the Lewis structures of these possible resonance structures, and use formal charges to predict which makes the greatest contribution to the resonance hybrid.
Several Lewis structures can be written for perbromate ion, BrO4-, the central Br with all single Br-O bonds, or with one, two, or three Br=O double bonds. Draw the Lewis structures of these possible resonance structures, and use formal charges to predict which makes the greatest contribution to the resonance hybrid.
Verified Answer
Kenneth DuqueLv10
26 Nov 2020
13 Dec 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.