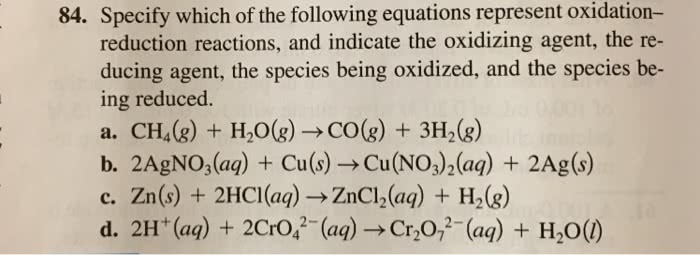1
answer
0
watching
504
views
6 Oct 2020
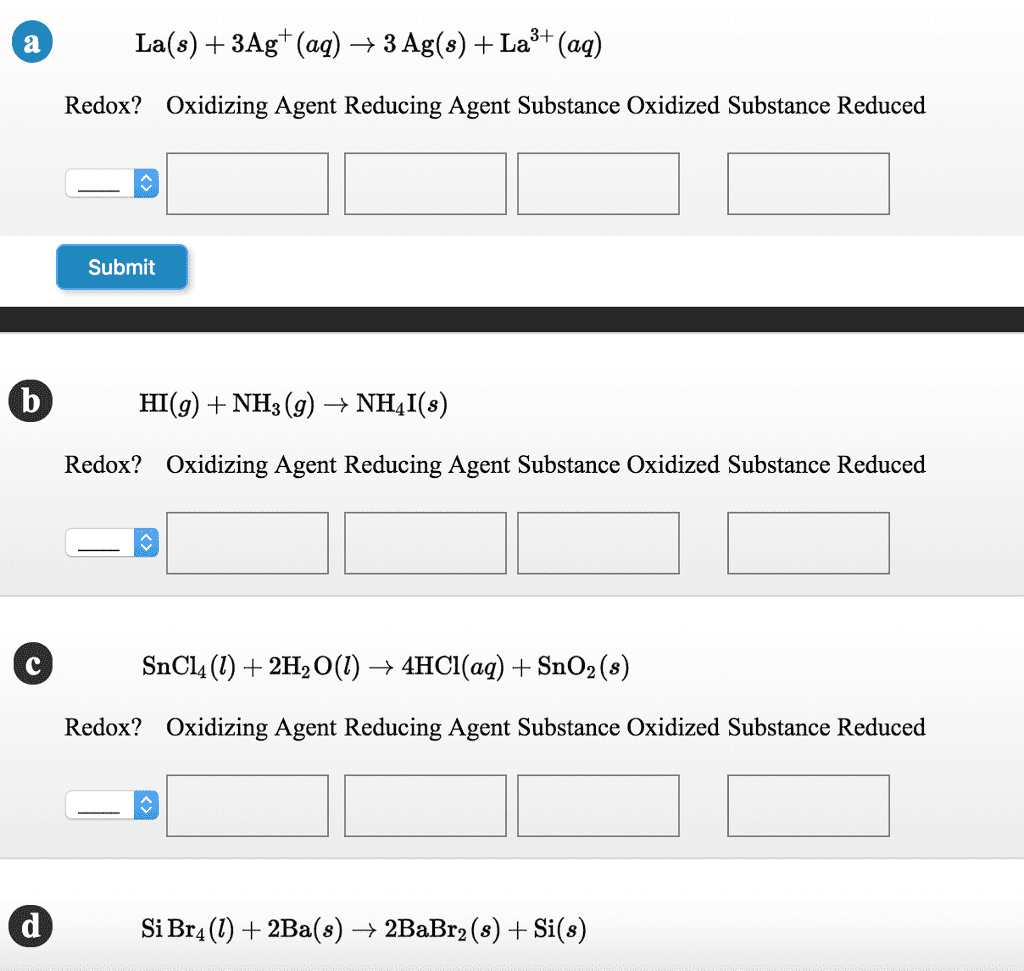
Specify which of the following are oxidation-reduction reactions, and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
- Cu (s) + 2Ag+ (aq) → 2Ag (s) + Cu2+ (aq)
- HCl (g) + NH3 (g) → NH4Cl (s)
- SiCl4 (l) + 2H2O (l) → 4HCl (aq) + SiO2 (s)
- SiCl4 (l) + 2Mg (s) → 2MgCl2 (s) + Si (s)
- Al(OH)4− (aq) → AlO2− (aq) + 2H2O (l)
Specify which of the following are oxidation-reduction reactions, and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
- Cu (s) + 2Ag+ (aq) → 2Ag (s) + Cu2+ (aq)
- HCl (g) + NH3 (g) → NH4Cl (s)
- SiCl4 (l) + 2H2O (l) → 4HCl (aq) + SiO2 (s)
- SiCl4 (l) + 2Mg (s) → 2MgCl2 (s) + Si (s)
- Al(OH)4− (aq) → AlO2− (aq) + 2H2O (l)
Robert KubaraLv10
25 Nov 2020