1
answer
0
watching
249
views
6 Oct 2020
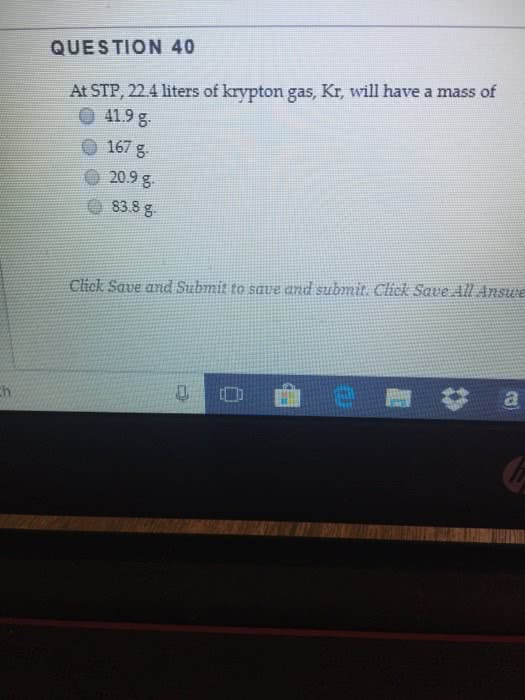
Given 3.00 moles of krypton gas, Kr (g), how many liters will this sample occupy at STP?
a. 11.2
b. 22.4
c. 44.8
d. 67.2
Given 3.00 moles of krypton gas, Kr (g), how many liters will this sample occupy at STP?
a. 11.2
b. 22.4
c. 44.8
d. 67.2
Christian TusoLv10
25 Nov 2020