1
answer
0
watching
142
views
6 Oct 2020
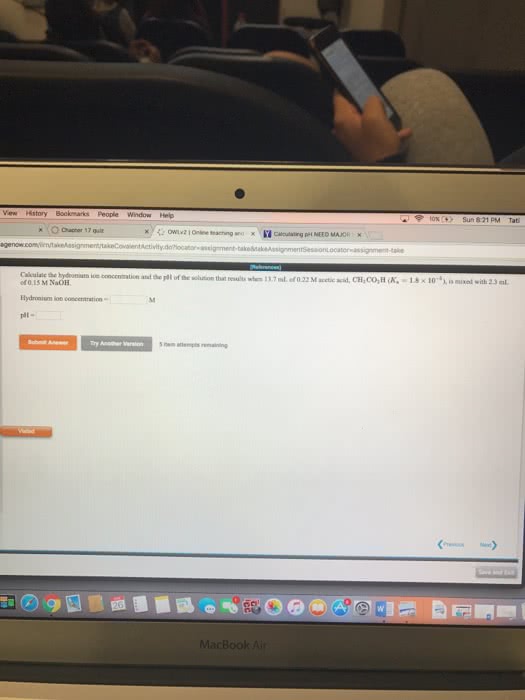
Calculate the hydronium ion concentration and pH of the solution that results when 22.0 mL of 0.15 M acetic acid, CH3COOH, is mixed with 22.0 mL of 0.15 M NaOH.
Calculate the hydronium ion concentration and pH of the solution that results when 22.0 mL of 0.15 M acetic acid, CH3COOH, is mixed with 22.0 mL of 0.15 M NaOH.
Patrick SuarezLv10
18 Nov 2020