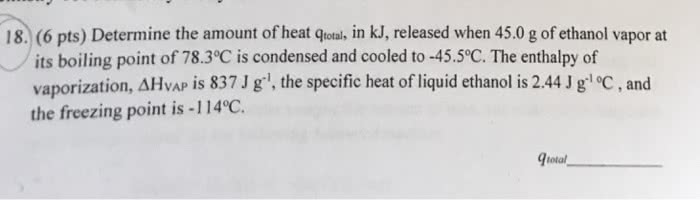1
answer
0
watching
237
views
6 Oct 2020
Ethanol, C2H8OH, boils at 78.29 °C. How much energy, in joules, is required to raise the temperature of 1.00 kg of ethanol from 20.0 °C to the boiling point and then change the liquid to vapor at that temperature? (The specific heat of ethanol is 2.44 J/g•K, and its enthalpy of vaporization is 855 J/g)
Ethanol, C2H8OH, boils at 78.29 °C. How much energy, in joules, is required to raise the temperature of 1.00 kg of ethanol from 20.0 °C to the boiling point and then change the liquid to vapor at that temperature? (The specific heat of ethanol is 2.44 J/g•K, and its enthalpy of vaporization is 855 J/g)
Christian TusoLv10
12 Nov 2020