1
answer
1
watching
205
views
6 Oct 2020
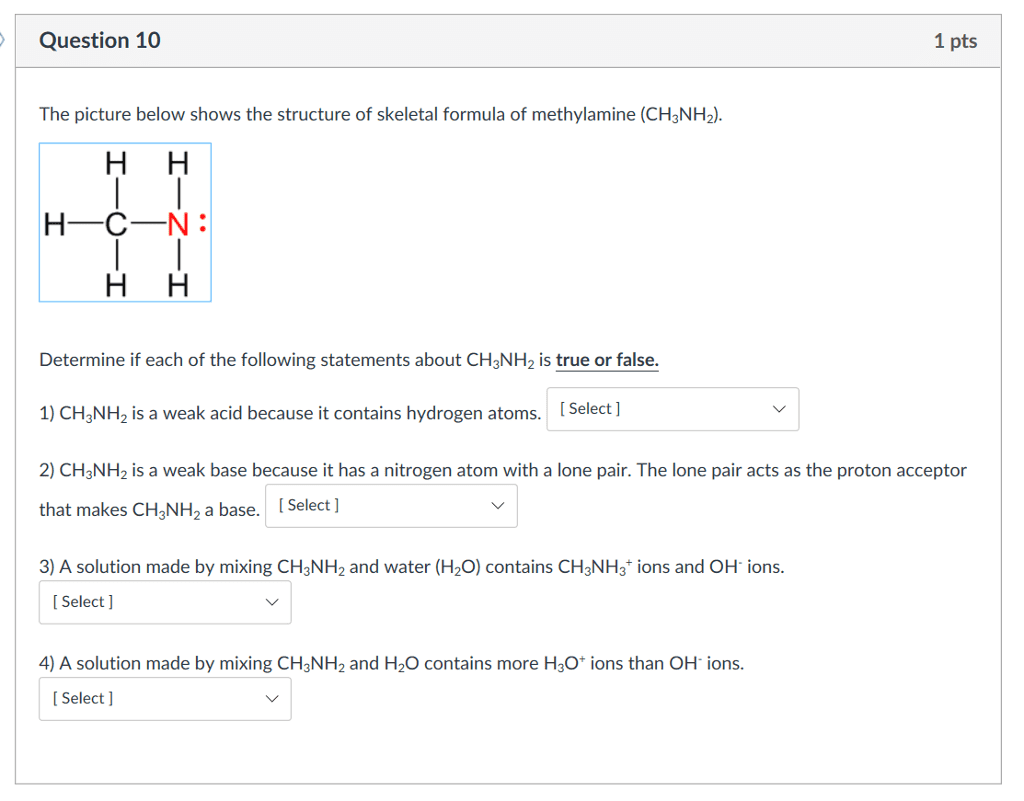
The weak base methylamine, CH3NH2, has Kb = 4.2 ×10−4. It reacts with water according to the equation.
CH3NH2 (aq) + H2O (l) ⇌ CH3NH3+ (aq) + OH− (aq)
Calculate the equilibrium hydroxide ion concentration in a 0.25 M solution of the base. What are the pH and pOH of the solution?
The weak base methylamine, CH3NH2, has Kb = 4.2 ×10−4. It reacts with water according to the equation.
CH3NH2 (aq) + H2O (l) ⇌ CH3NH3+ (aq) + OH− (aq)
Calculate the equilibrium hydroxide ion concentration in a 0.25 M solution of the base. What are the pH and pOH of the solution?
Robert KubaraLv10
11 Nov 2020