1
answer
0
watching
215
views
6 Oct 2020
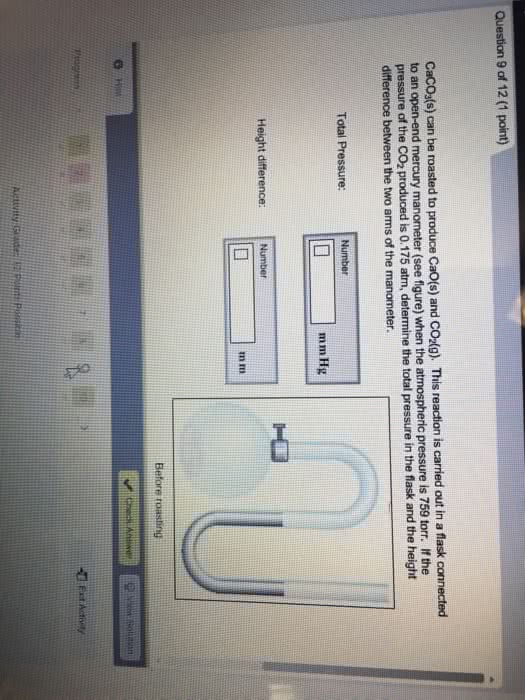
For each of the following situations where a gas is contained in a flask, calculate the pressure in the flask in torr, atm and pascal.

c. Calculate the pressure in the flask in part a and b (in torr) if the atmospheric pressure is 635 torr
For each of the following situations where a gas is contained in a flask, calculate the pressure in the flask in torr, atm and pascal.

c. Calculate the pressure in the flask in part a and b (in torr) if the atmospheric pressure is 635 torr
Allen DinoLv9
18 Nov 2020