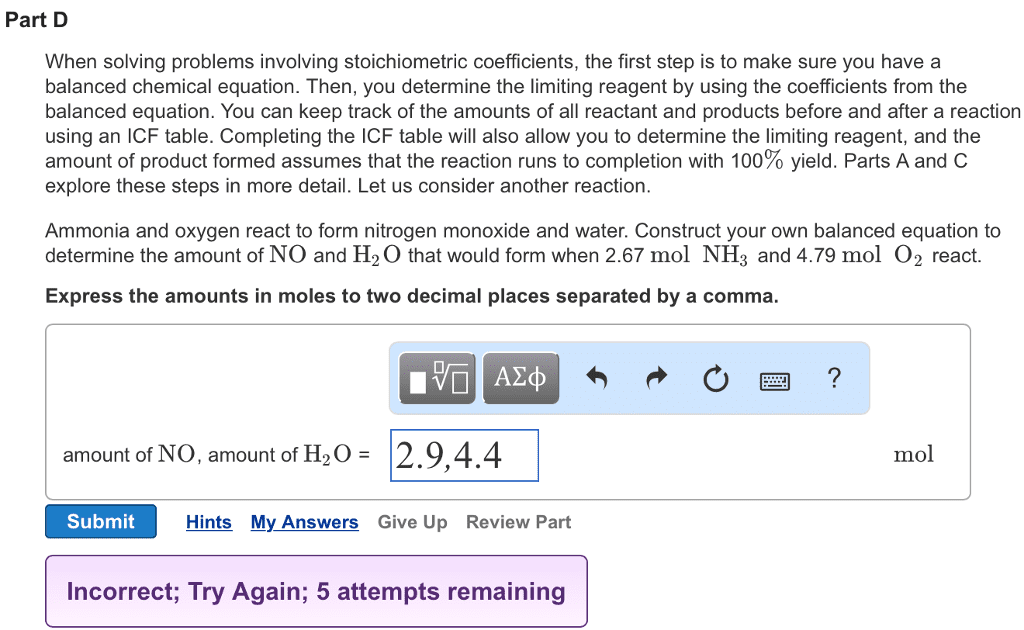
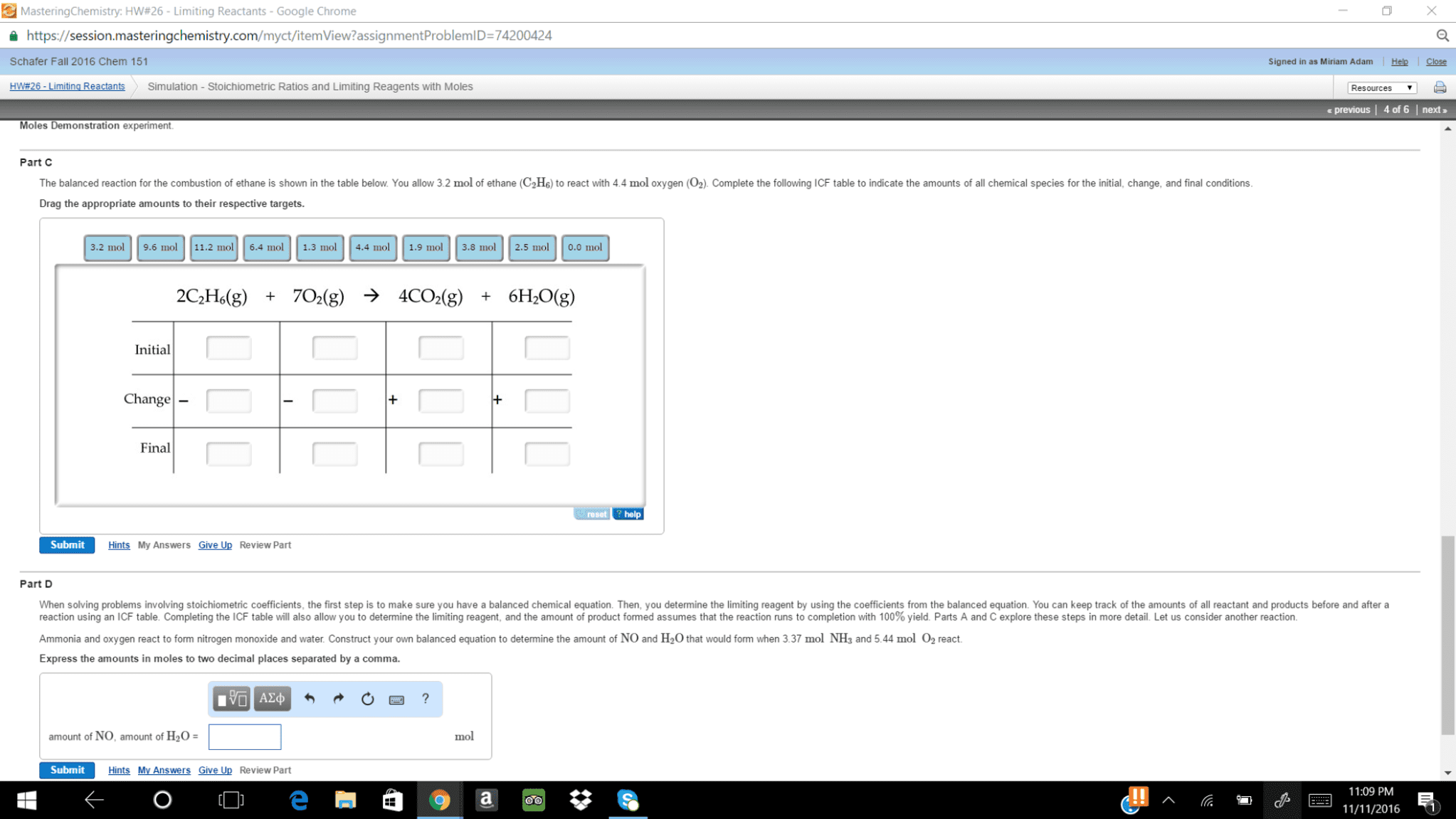
When solving problems involving stoichiometric coefficients, the first step is to make sure you have a balanced chemical equation. Then, you determine the limiting reagent by using the coefficients from the balanced equation. You can keep track of the amounts of all reactant and products before and after a reaction using an ICF table (as shown in the Simulation). Completing the ICF table will also allow you to determine the limiting reagent, and the amount of product formed is based on assuming that the reaction runs to completion with 100% yield. Parts A and C explore these steps in more detail. Let us consider another reaction. Ammonia and oxygen react to form nitrogen monoxide and water. Construct your own balanced equation to determine the amount of NO and H2O that would form when 3.49 mol NH3 and 5.86 mol O2 react.
When solving problems involving stoichiometric coefficients, the first step is to make sure you have a balanced chemical equation. Then, you determine the limiting reagent by using the coefficients from the balanced equation. You can keep track of the amounts of all reactant and products before and after a reaction using an ICF table (as shown in the Simulation). Completing the ICF table will also allow you to determine the limiting reagent, and the amount of product formed is based on assuming that the reaction runs to completion with 100% yield. Parts A and C explore these steps in more detail. Let us consider another reaction. Ammonia and oxygen react to form nitrogen monoxide and water. Construct your own balanced equation to determine the amount of NO and H2O that would form when 3.49 mol NH3 and 5.86 mol O2 react.