1
answer
0
watching
427
views
28 Nov 2020
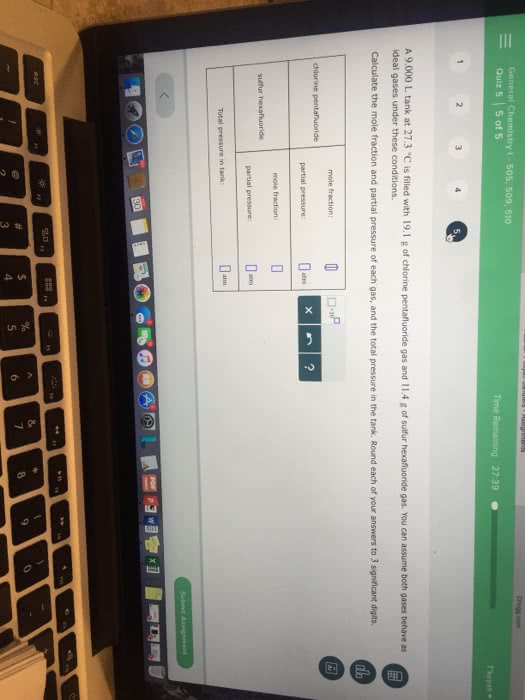
A 9.00 L tank at 16.4°C is filled with 15.8 g of dinitrogen difluoride gas and 2.60 g of chlorine pentafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits.
A 9.00 L tank at 16.4°C is filled with 15.8 g of dinitrogen difluoride gas and 2.60 g of chlorine pentafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits.
Verified Answer
Alyssa Mae RemarimLv10
6 Jan 2021
16 Jan 2021
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.