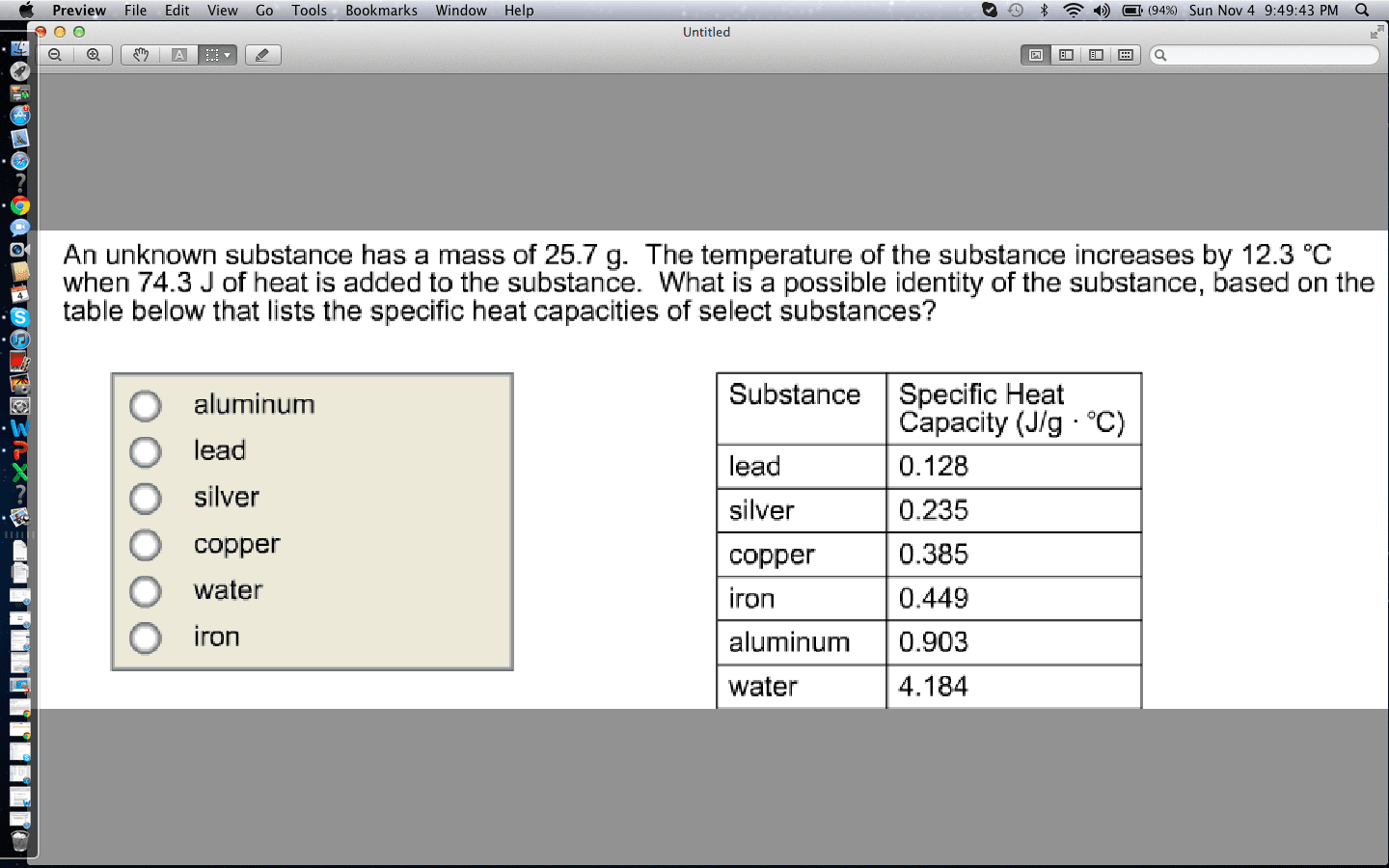
2
answers
0
watching
192
views
28 Nov 2020
A 51 g aluminum block initially at 27.5 °C absorbs 715 J of heat. What is the final temperature of the aluminum? Express your answers using one decimal place and include the appropriate units.
Substance
Specific heat capacity, c (J/g °C) at 298 K
Elements
Lead
0.128
Gold
0.128
Silver
0.235
Copper
0.385
Iron
0.449
Aluminum
0.903
A 51 g aluminum block initially at 27.5 °C absorbs 715 J of heat. What is the final temperature of the aluminum? Express your answers using one decimal place and include the appropriate units.
| Substance | Specific heat capacity, c (J/g °C) at 298 K |
| Elements | |
| Lead | 0.128 |
| Gold | 0.128 |
| Silver | 0.235 |
| Copper | 0.385 |
| Iron | 0.449 |
| Aluminum | 0.903 |
Patrick SuarezLv10
6 Jan 2021
Already have an account? Log in