1
answer
0
watching
702
views
28 Nov 2020
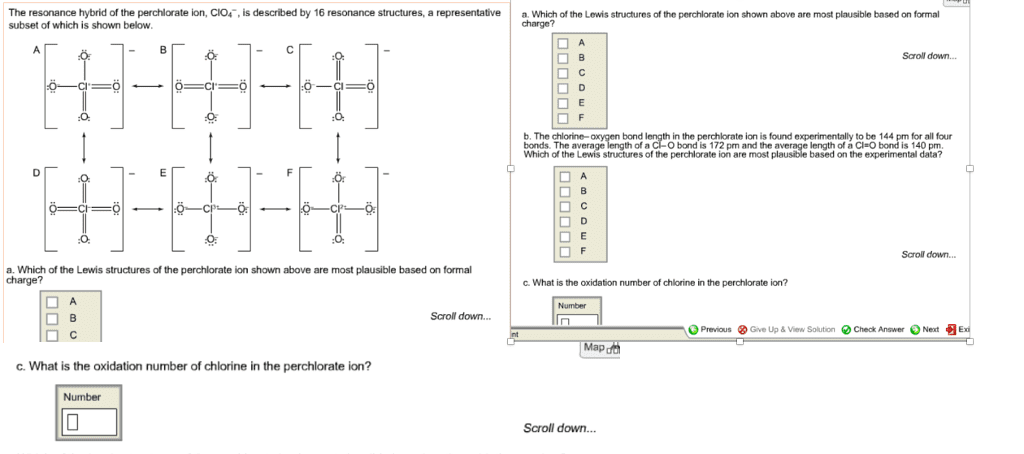
(a) Determine the formal charge on the chlorine atom in the perchlorate ion, ClO4-, and the chlorate ion, ClO3-.
(b) What are the oxidation numbers of chlorine in ClO4- and in ClO3-?
(a) Determine the formal charge on the chlorine atom in the perchlorate ion, ClO4-, and the chlorate ion, ClO3-.
(b) What are the oxidation numbers of chlorine in ClO4- and in ClO3-?
Bryllant BaluyutLv10
26 Jan 2021