2
answers
1
watching
456
views
tealdog287Lv1
28 Feb 2021
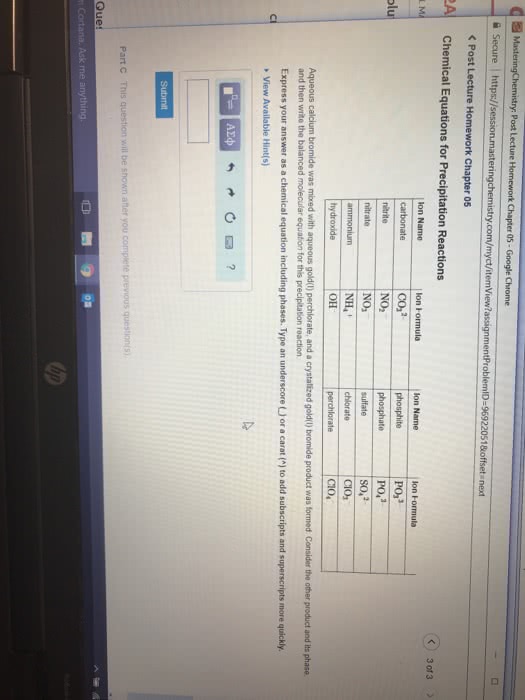
Below is a list of some common polyatomic ions for your reference.
Ion Name
Ion Formula
Ion Name
Ion Formula
carbonate
CO32−CO32−
phosphite
PO33−PO33−
nitrite
NO2−NO2−
phosphate
PO43−PO43−
nitrate
NO3−NO3−
sulfate
SO42−SO42−
ammonium
NH4+NH4+
chlorate
ClO3−ClO3−
hydroxide
OH−OH−
perchlorate
ClO4−ClO4−
Aqueous lithium sulfate was mixed with aqueous strontium chlorate, and a crystallized strontium sulfate product was formed. A crystalized product is a solid. The other product, lithium chlorate, remains dissolved in solution. Consider the other product and its phase, and then write the balanced molecular equation for this precipitation reaction.
Express your answer as a chemical equation including phases. Type an underscore (_) or a carat (^) to add subscripts and superscripts more quickly.
Below is a list of some common polyatomic ions for your reference.
| Ion Name | Ion Formula | Ion Name | Ion Formula |
| carbonate | CO32−CO32− | phosphite | PO33−PO33− |
| nitrite | NO2−NO2− | phosphate | PO43−PO43− |
| nitrate | NO3−NO3− | sulfate | SO42−SO42− |
| ammonium | NH4+NH4+ | chlorate | ClO3−ClO3− |
| hydroxide | OH−OH− | perchlorate | ClO4−ClO4− |
Aqueous lithium sulfate was mixed with aqueous strontium chlorate, and a crystallized strontium sulfate product was formed. A crystalized product is a solid. The other product, lithium chlorate, remains dissolved in solution. Consider the other product and its phase, and then write the balanced molecular equation for this precipitation reaction.
Express your answer as a chemical equation including phases. Type an underscore (_) or a carat (^) to add subscripts and superscripts more quickly.
Read by 1 person
John Edward CayasLv10
28 Feb 2021
Already have an account? Log in