2
answers
1
watching
321
views
18 Mar 2021
Consider the energy profile diagram below. (The profile is not drawn to scale).
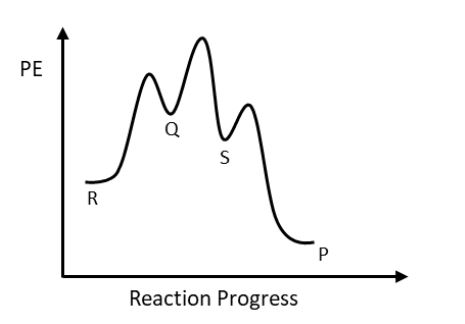
A: Consider the following activation energies for the overall reaction R → P.
Efora= 55 kJ mol−1
Ereva= 106 kJ mol−1
What is the enthalpy change for the overall reaction R → P?
ΔH= kJ mol−1
B: Which of the following statements regarding the overall reaction R → P is/are incorrect? Select all that apply.
Three activated complexes are formed in this reaction
Step S→P has a smaller rate constant than step P→S
Step Q→S is the rate-determining step
The step R→Q has the largest rate constant
ΔrH is positive
Species S is an intermediate
Consider the energy profile diagram below. (The profile is not drawn to scale).
A: Consider the following activation energies for the overall reaction R → P.
Efora= 55 kJ mol−1
Ereva= 106 kJ mol−1
What is the enthalpy change for the overall reaction R → P?
ΔH= kJ mol−1
B: Which of the following statements regarding the overall reaction R → P is/are incorrect? Select all that apply.
|
Three activated complexes are formed in this reaction |
||
|
Step S→P has a smaller rate constant than step P→S |
||
|
Step Q→S is the rate-determining step |
||
|
The step R→Q has the largest rate constant |
||
|
ΔrH is positive |
||
|
Species S is an intermediate |
irullandi226Lv1
30 Apr 2023
Anika ArnaldoLv10
18 Mar 2021
Already have an account? Log in


