3
answers
0
watching
16
views
19 Nov 2019
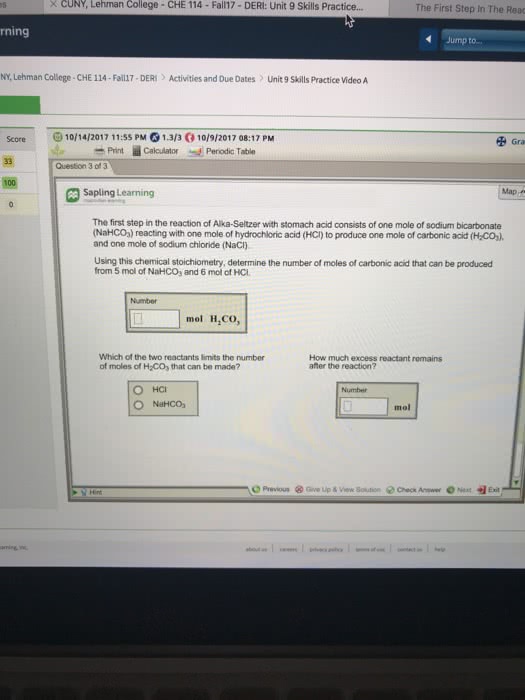
The first step in the reaction of Alka-Seltzer with stomach acid consists of one mole of sodium bicarbonate (NaHCO3) reacting with one mole of hydrochloric acid (HCl) to produce one mole of carbonic acid (H2CO3), and one mole of sodium chloride (NaCl). Using this chemical stoichiometry, determine the number of moles of carbonic acid that can be produced from 2 mol of NaHCO3 and 7 mol of HCl.
The first step in the reaction of Alka-Seltzer with stomach acid consists of one mole of sodium bicarbonate (NaHCO3) reacting with one mole of hydrochloric acid (HCl) to produce one mole of carbonic acid (H2CO3), and one mole of sodium chloride (NaCl). Using this chemical stoichiometry, determine the number of moles of carbonic acid that can be produced from 2 mol of NaHCO3 and 7 mol of HCl.
5 Jun 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
2 Jun 2023
Get unlimited access
Already have an account? Log in
Jarrod RobelLv2
22 Mar 2019
Get unlimited access
Already have an account? Log in