2
answers
0
watching
4
views
19 Nov 2019
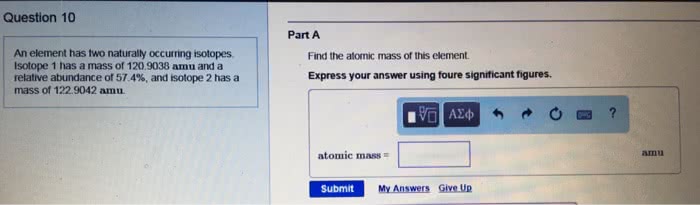
the element lithium has two naturally occurring isotopes. one of these has a mass of 6.0151 amu and a natural abundance of 7.49%. a second isotope has a mass of 7.0160amu and a natural abundance of 92.51%. calculate the atomic mass of lithium
the element lithium has two naturally occurring isotopes. one of these has a mass of 6.0151 amu and a natural abundance of 7.49%. a second isotope has a mass of 7.0160amu and a natural abundance of 92.51%. calculate the atomic mass of lithium
28 Jun
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Collen VonLv2
7 Feb 2019
Get unlimited access
Already have an account? Log in