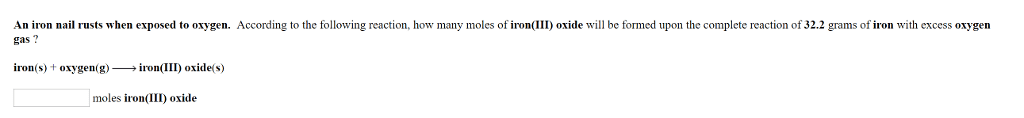1
answer
0
watching
27
views
23 Nov 2019
Iron reacts with oxygen to produce iron (III) oxide.
4Fe(s) + 3O2(g) -> 2Fe2O3(s)
If 3.2 molecules of Fe react with excess O2, how many moles ofFe2O3 can be formed?
Iron reacts with oxygen to produce iron (III) oxide.
4Fe(s) + 3O2(g) -> 2Fe2O3(s)
If 3.2 molecules of Fe react with excess O2, how many moles ofFe2O3 can be formed?
saubhagyaLv2
6 May 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in