2
answers
0
watching
24
views
23 Nov 2019
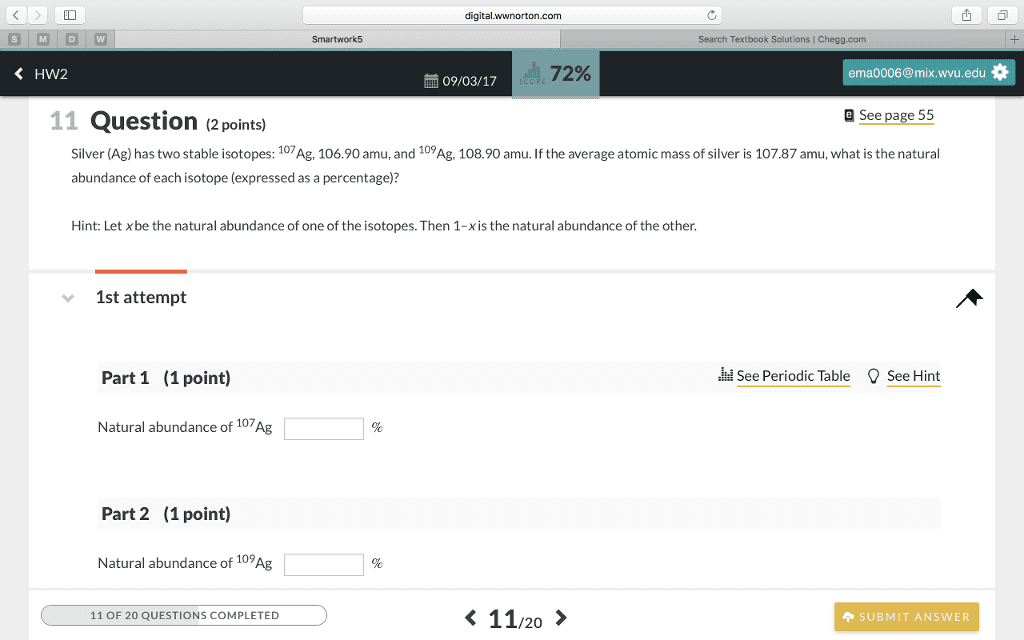
There are two stable isotopes of copper. Their masses are 62.9296amu and 64.9278 amu. If the average atomic mass of copper is 63.55,what is the natural abundance of the lighter isotope?
There are two stable isotopes of copper. Their masses are 62.9296amu and 64.9278 amu. If the average atomic mass of copper is 63.55,what is the natural abundance of the lighter isotope?
sunieltanejaLv3
24 Jun 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Bunny GreenfelderLv2
6 Oct 2019
Get unlimited access
Already have an account? Log in