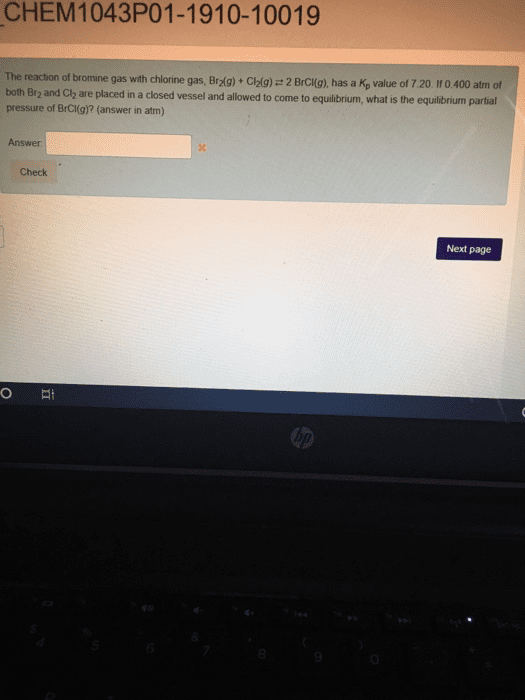
2
answers
0
watching
14
views
23 Nov 2019
A sample of Cl2 with a pressure of 1.96 atm and a volume of 228 mL is allowed to react with excess Br2 at 114 °C. Br2 (g) + Cl2 (g) 2 BrCl (g) Calculate the pressure of the BrCl produced in the reaction if it is transferred to a 2.31-L flask and cooled to 36 °C. ___ atm
A sample of Cl2 with a pressure of 1.96 atm and a volume of 228 mL is allowed to react with excess Br2 at 114 °C. Br2 (g) + Cl2 (g) 2 BrCl (g) Calculate the pressure of the BrCl produced in the reaction if it is transferred to a 2.31-L flask and cooled to 36 °C. ___ atm
thanhmai1309Lv9
31 Aug 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Lelia LubowitzLv2
2 Oct 2019
Get unlimited access
Already have an account? Log in